Biotechnology
Innovent Announces NMPA Acceptance of a Supplemental New Drug Application for Sintilimab in Combination with Chemotherapy as First-Line Therapy for Esophageal Squamous Cell Carcinoma (ESCC)
SAN FRANCISCO and SUZHOU, China, Sept. 23, 2021 /PRNewswire/ -- Innovent Biologics, Inc. (Innovent) (HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures and commercializes high quality medicines for the treatment of cancer, metabolic, autoimmune and other major disea...
MedAlliance announces completion of enrollment in Japanese SELUTION SLR™ Study
GENEVA, Sept. 23, 2021 /PRNewswire/ -- MedAlliance, with its Japanese partner MDK Medical, has completed enrollment in the clinical study of its novel Sirolimus Drug Eluting Balloon (DEB), SELUTION SLR™, for the treatment of peripheral arterial disease (PAD). This follows the acceptance of a Clin...
INOVIO Receives Regulatory Authorization to Conduct Phase 3 Efficacy Trial of its COVID-19 DNA Vaccine Candidate, INO-4800, in Mexico
News follows recent regulatory authorizations to proceed with the trial in Brazil and the Philippines INOVIO and partner Advaccine are collaborating on the global Phase 3 trial in regions underserved by COVID-19 vaccines; to focus onLatin America, Asia, and Africa PLYMOUTH MEETING, Pa., Sept. 2...
Clover's COVID-19 Vaccine Candidate Demonstrates 79% Efficacy Against Delta in Global Phase 2/3 SPECTRA Trial Dominated by Variants of Concern and Interest
* Trial enrolled over 30,000 adult & elderly participants across 4 continents; 100% of SARS-CoV-2 strains observed in efficacy analysis were variants (Delta was predominant strain) * Primary and secondary efficacy endpoints were successfully met * 100% efficacy against severe COVID-19 & hosp...
Samsung Biologics showcases its newest CDO process platform, S-Cellerate™, at BPI 2021, offering expedited timeline to IND and BLA
INCHEON, South Korea, Sept. 22, 2021 /PRNewswire/ -- Samsung Biologics, a global contract development and manufacturing organization (CDMO), introduced its proprietary technology platform, S-CellerateTM, at the BioProcess International Conference (BPI 2021) to offer clients an integrated and seam...
Asieris Appoints Dr. Alice Chen as Vice President, Discovery Biology and Head of Translational Research
SHANGHAI, Sept. 22, 2021 /PRNewswire/ -- Asieris Pharmaceuticals announced the appointment of Dr.Alice Chen as Vice President, Discovery Biology and Head of Translational Research with effect fromSeptember 20, 2021. She will report to Dr.Kevin Pan, Founder and CEO of Asieris. Alice has over 20 ...
New Therapy to Treat Advanced Small Cell Lung Cancer ZEPZELCA® (lurbinectedin) Approved in Singapore
Highlights: * ZEPZELCA® (lurbinectedin) is the first new therapy approved in Singapore to treat 2L metastatic small cell lung cancer (SCLC) in 20 years * ZEPZELCA provisional approval represents an important advance for adult patients whose metastatic SCLC has progressed on or after platinum-...
LSO Medical Announces CE Approval of Next-Gen LUMESEAL Laser for Treatment of Varicose Vein.
LILLE, France, Sept. 22, 2021 /PRNewswire/ -- LSO MEDICAL SAS, international leader in vascular laser therapies, announced today the CE Approval of its newest generation EVLT SnakeBack® assisted system, the LUMESEAL® laser platform. Designed to enhance ease-of-use and to provide greater precision...
Origin Agritech and China Agricultural University to Collaborate on Research of Molecular Design of Corn Using CRISPR
BEIJING, Sept. 21, 2021 /PRNewswire/ -- Origin Agritech Ltd. (NASDAQ: SEED) (the "Company" or "Origin"), an agriculture technology company, today announced that it has entered into collaboration agreement with China Agricultural University for the research of molecular design in corn. One of the...
Novavax to Participate in Fireside Chat at Devex @ UNGA 76
GAITHERSBURG, Md., Sept. 21, 2021 /PRNewswire/ -- Novavax, Inc. (Nasdaq: NVAX), a biotechnology company developing and commercializing next-generation vaccines for serious infectious diseases, today announced thatJohn J. Trizzino, Executive Vice President, Chief Commercial Officer and Chief Busin...
RhoVac announces the completion of patient recruitment for the BRaVac clinical phase IIb study of RV001 in prostate cancer
STOCKHOLM, Sept. 21, 2021 /PRNewswire/ -- RhoVac AB ("RhoVac"), a Swedish cancer immunotherapy company, announces today onSep 20th , 2021, that its clinical phase IIb study of lead drug candidate RV001 in prostate cancer, BRaVac, is deemed fully recruited and that no additional patients will be ...
Alterity Announces Presentation of Biomarker Data at the International Parkinson and Movement Disorder Society Congress 2021
MELBOURNE, Australia and SAN FRANCISCO, Sept. 20, 2021 /PRNewswire/ -- Alterity Therapeutics (ASX: ATH, NASDAQ: ATHE) ("Alterity" or "the Company"), a biotechnology company dedicated to developing disease modifying treatments for neurodegenerative conditions, today announced that data was present...
PharmAbcine to Participate in BIO-Europe 2021
DAEJEON, South Korea, Sept. 20, 2021 /PRNewswire/ -- PharmAbcine Inc. (KOSDAQ: 208340ks), a clinical-stage biotech company focusing on the development of next generation antibody therapeutics, announced today that the Company will virtually participate in the 27th annual BIO-Europe which will tak...
Bionomics Prepares BNC210 for Start of Phase 2 Acute Treatment of Social Anxiety Disorder Trial
* Rapid oral absorption of BNC210 novel tablet formulation potentially well-suited for acute treatment of anxiety in patients with Social Anxiety Disorder * Phase 2 clinical trial on target to start by end of 2021 and expected to read out topline data by end of 2022 ADELAIDE, Australia, Sept....
Terumo celebrates 100-year anniversary in Asia with an eye to the future
SINGAPORE, Sept. 20, 2021 /PRNewswire/ -- Terumo, a global leader in medical technology, celebrates its centenary inAsia. From humble beginnings as a thermometer manufacturer in 1921, and the first company in 1963 to create disposable syringes inJapan, the company has been providing medical solut...
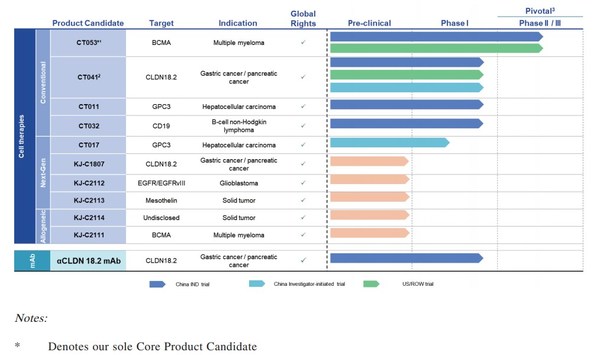
Appealing Data of CARsgen Therapeutics' CAR-T (CT041) in Advanced Gastric Cancer Presented at ESMO
SHANGHAI, Sept. 19, 2021 /PRNewswire/ -- On September 19, 2021, CARsgen Therapeutics (stock code: 2171.HK) disclosed the latest progress of the investigator-initiated trial (IIT) of Claudin18.2 (CLDN18.2) CAR-T (CT041) for the treatment of digestive system tumors. Results of this trial have been ...
Illumina reaffirms commitment to the National Project of Bio Big Data in Korea
MELBOURNE, Australia, Sept. 20, 2021 /PRNewswire/ -- Illumina, Inc. (NASDAQ: ILMN) today announced that the National Project of Bio Big Data in Korea has selected Illumina technology for the second pilot project in their effort to work towards establishing a national digital library on health and...
2021 CSCO | Henlius will Release Four Oral Presentations of Two Upcoming Commercial Products: Novel anti-PD-1 mAb Serplulimab and Bevacizumab Biosimilar
SHANGHAI, Sept. 18, 2021 /PRNewswire/ -- The 24th Annual Meeting of Chinese Society of Clinical Oncology (CSCO) will be held online and in-person from 25th to 29th September 2021. In this meeting, Henlius will release 4 study results of 2 products to be commercialized, the novel anti-PD-1 mAb ser...
Innovent Releases Interim Analysis Results of Sintilimab plus Chemotherapy for the First-Line Treatment of Esophageal Squamous Cell Carcinoma in the Phase 3 ORIENT-15 Study at ESMO Congress 2021
SAN FRANCISCO and SUZHOU, China, Sept. 18, 2021 /PRNewswire/ -- Innovent Biologics, Inc. ("Innovent") (HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures and commercializes high quality medicines for the treatment of cancer, metabolic, autoimmune and other major dis...
The NDA of Henlius Novel Anti-PD-1 mAb Serplulimab for First-Line Treatment of sqNSCLC Accepted by China's NMPA, Phase 3 MRCT Met its Primary Endpoint
SHANGHAI, Sept. 17, 2021 /PRNewswire/ -- Shanghai Henlius Biotech, Inc. (2696.HK) announced that the New Drug Application (NDA) of Serplulimab Injection (HLX10), a novel anti-PD-1 monoclonal antibody (mAb) independently developed by the company, in combination with carboplatin and albumin-bound ...
Week's Top Stories
Most Reposted
Visa partners with Laufey to spread the magic of travel in Asia Pacific; to be Official Payment Partner for Laufey: A Matter of Time Tour
[Picked up by 310 media titles]
2026-03-04 12:35TelkomMetra Streamlines Business Portfolio, AdMedika Group Poised to Enter a New Growth Phase on Regional Level Under Fullerton Health
[Picked up by 295 media titles]
2026-03-09 21:36Progress Software Highlights Load Balancing as a Strategic Imperative for APAC Digital Infrastructure Resilience
[Picked up by 289 media titles]
2026-03-10 09:00COL and NASDAQ-Listed BeLive Holdings Unveil World's First "Microdrama in a Box" in Headline Hong Kong FILMART 2026 Launch
[Picked up by 287 media titles]
2026-03-05 17:14Gene Solutions Announces Strategic Entry into Türkiye with Robust Real-World Evidence at the 2nd International Hereditary Cancers Congress 2026
[Picked up by 267 media titles]
2026-03-04 10:00