Biotechnology
Prenetics and New World Development Launch Circle HealthPod, the World's Most Advanced At-Home Covid-19 test
* Circle HealthPod is a Rapid Detection System for all infectious diseases, starting with Covid-19 and will include Flu and STDs in the future * Technology developed at University of Oxford, with PCR quality results in 20 minutes * Circle HealthPod, having received CE-IVD marking, is a reusa...
Insilico Medicine Announces an AI-driven PROTAC R&D Collaboration with Arvinas
HONG KONG, Aug. 9, 2021 /PRNewswire/ -- Insilico Medicine is proud to announce an R&D collaboration with Arvinas (NASDAQ: ARVN). This strategic partnership will involve Insilico Medicine's AI-augmented platforms and close cooperation between Arvinas' and Insilico Medicine's scientists on novel PR...
YishengBio Announces PIKA recombinant COVID-19 vaccine received Phase I clinical trial IND clearance from New Zealand
BEIJING, Aug. 9, 2021 /PRNewswire/ -- YishengBio Co., Ltd ("YishengBio") today announced that it received IND clearance for PIKA recombinant COVID-19 vaccine (CHO cell, S Protein) to conduct Phase I clinical trial from New Zealand Medicines and Medical Devices Safety Authority ("MEDSAFE"), which ...
Life Sciences' Contribution to the US Digital Therapeutics Market to Grow the Fastest amongst all End Users
The US digital therapeutics market is estimated to witness more than three-and-a-half-fold growth reaching$4.54 billion by 2025, at a CAGR of 29.8%, finds Frost & Sullivan SANTA CLARA, Calif., Aug. 9, 2021 /PRNewswire/ -- Frost & Sullivan's recent analysis finds that digital therapeutics (DTx) i...
Thailand's Jan-June Investment Applications Reach USD12 billion, as FDI Quadruple, E&E and Medical Sectors Continue to Expand, BOI says
BANGKOK, Aug. 9, 2021 /PRNewswire/ -- In the first six months of 2021, Thailand's investment applications increased 14% from the year earlier period in terms of the number of projects, and 158% in combined value, led by increasing foreign direct investment (FDI) applications, sustained growth in ...
Antengene and MindRank AI Enter into Collaboration to Advance the Development of Difficult-to-Drug Molecular Targets
SHANGHAI and HONG KONG, Aug. 9, 2021 /PRNewswire/ -- Antengene Corporation Limited ("Antengene", SEHK: 6996.HK), a leading innovative biopharmaceutical company dedicated to discovering, developing and commercializing global first-in-class and/or best-in-class therapeutics in hematology and oncolo...
FibroBiologics Announces Inaugural Board of Directors
Company Announces Formation of Board of Directors HOUSTON, Aug. 6, 2021 /PRNewswire/ -- FibroBiologics, a clinical stage company developing fibroblast-based therapeutic solutions for chronic diseases, announced today the formation of its Board of Directors. Leading this group is Chairman,Pete ...
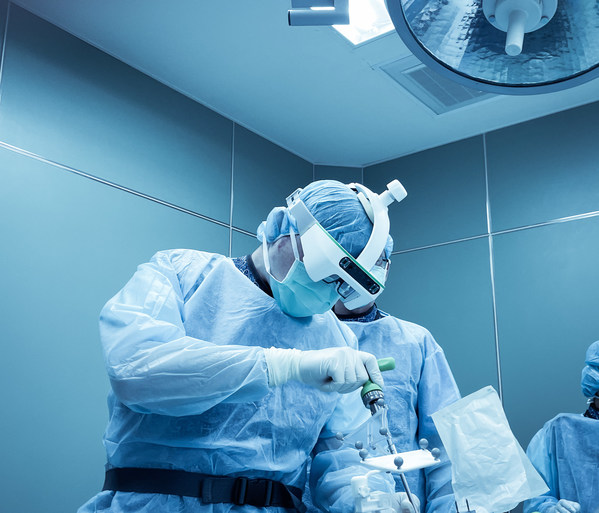
SURGLASSES' SPINE NAVIGATION SYSTEM CADUCEUS S - An innovation greatly influencing minimally invasive surgeries
TAICHUNG, Aug. 6, 2021 /PRNewswire/ -- Taiwan Main Orthopaedic Biotechnology Co. Ltd (SURGLASSES),Taiwan, a company striving to bring a difference into the real time surgical procedure after the introduction of the first AR assisted Head Mounted Display Foresee-X, is now coming up with a new prod...
Updated Overall Survival Data and Biomarker Results from Sintilimab ORIENT-11 Study in First-Line Nonsquamous Non-Small Cell Lung Cancer Published in the Journal of Thoracic Oncology
SAN FRANCISCO and SUZHOU, China, Aug. 6, 2021 /PRNewswire/ -- Innovent Biologics, Inc. ("Innovent") (HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures and commercializes high-quality medicines for the treatment of oncology, metabolic, autoimmune and other major dis...
Novavax and Serum Institute of India Announce Submission to Regulatory Agencies in India, Indonesia, Philippines for Emergency Use Authorization of Novavax' Recombinant Nanoparticle COVID-19 Vaccine
- Novavax and Serum Institute of India file regulatory submissions to the Drugs Controller General ofIndia and to regulatory agencies in Indonesia and Philippines for Novavax' recombinant nanoparticle COVID-19 vaccine - Filing for World Health Organization Emergency Use Listing expected in Augus...
Advanced Instruments to Acquire Solentim, a Trusted Global Leader in Cell Imaging for Cell Line Development and Clone Characterization
NORWOOD, Mass., Aug. 6, 2021 /PRNewswire/ -- Advanced Instruments, a leading manufacturer of analytical instruments and services for bioprocessing, today announced the execution of a definitive agreement to acquire Solentim, a trusted global leader of solutions for the isolation and characterizat...
ArisGlobal Lauded by Frost & Sullivan for Enabling Life Sciences Companies to Accelerate R&D with Its LifeSphere® Platform
LifeSphere® ably supports clients by expediting decision-making, maintaining compliance, and streamlining collaborations among cross-functional teams SANTA CLARA, Calif., Aug. 5, 2021 /PRNewswire/ -- Based on its recent analysis of the North American clinical development and pharmacovigilance so...
Inmagene and Kissei Enter into an Exclusive License Agreement for Fostamatinib in China
SHANGHAI and SAN DIEGO and NAGANO, Japan, Aug. 5, 2021 /PRNewswire/ -- Inmagene Biopharmaceuticals ("Inmagene") and Kissei Pharmaceutical Co., Ltd. ("Kissei") announced today that they have entered into an exclusive license agreement for the development and commercialization of fostamatinib in ma...
Everest Medicines Announces Approval of Clinical Trial Application by China NMPA for Phase 3 Trial of Xerava(TM) for Community-Acquired Bacterial Pneumonia
SHANGHAI, Aug. 5, 2021 /PRNewswire/ -- Everest Medicines
Wealth Dynamix Wins 'XCelent Functionality Award' in New Wealth Management CRM Report
LONDON, Aug. 5, 2021 /PRNewswire/ -- Wealth Dynamix
ABL Bio Receives IND Approval for Phase 1 Clinical Trial of ABL501, an anti-LAG-3/PD-L1 Bispecific Antibody
* South Korea's MFDS clears IND application for a Phase 1 study evaluating the safety and preliminary efficacy of ABL501 as a monotherapy for solid tumor treatment * ABL's third IND this year for bispecific antibody therapeutics SEONGNAM, South Korea, Aug. 4, 2021 /PRNewswire/ -- ABL Bio, Inc...
Novavax and European Commission Finalize Advance Purchase Agreement for up to 200 million doses of COVID-19 Vaccine
GAITHERSBURG, Md., Aug. 4, 2021 /PRNewswire/ -- Novavax, Inc. (Nasdaq: NVAX), a biotechnology company developing next-generation vaccines for serious infectious diseases, today announced that it has reached an agreement with the European Commission (EC) for the purchase of up to 200 million doses...
Alterity Therapeutics Announces New US Patent to Expand its Portfolio of Compounds for Neurodegenerative Diseases including Alzheimer's and Parkinson's
MELBOURNE, Australia, Aug. 4, 2021 /PRNewswire/ -- Alterity Therapeutics (ASX: ATH, NASDAQ: ATHE) ("Alterity" or "the Company"), a biotechnology company dedicated to developing disease modifying treatments for neurodegenerative conditions, today announced the United States Patent and Trademark Of...
Clarivate Acquires Bioinfogate, Reinforcing Position as Premier Provider of End-to-End Research Intelligence Solutions for Life Sciences
LONDON, Aug. 4, 2021 /PRNewswire/ -- Clarivate Plc (NYSE:CLVT), a global leader in providing trusted information and insights to accelerate the pace of innovation, today announced that it has acquired Bioinfogate, a leading provider of analytics solutions in the life sciences and producer of the ...
Varigard Technologies Proven to Prevent the Tactile Spread of SARS-CoV-2, B.1.617.2 - DELTA Variant
Varigard's protective Hand Sanitizing lotion prevents the tactical spread of SARS-CoV-2, B.1.617.2 for two hours and for over 20 hours with its organic Surface Spray FOLEY, Ala., Aug. 3, 2021 /PRNewswire/ -- Varigard and Synergy Laboratories have announced the test results of an advanced hand sa...
Week's Top Stories
Most Reposted
Visa and Trip.com Group Ink Strategic Collaboration, Enabling Consumers to Chase Their Passions Through Travel
[Picked up by 309 media titles]
2026-05-19 18:10Two-day Global Prosperity Summit 2026 officially opens
[Picked up by 308 media titles]
2026-05-19 23:59Two‑day Global Prosperity Summit 2026 concludes successfully
[Picked up by 303 media titles]
2026-05-21 22:13Hyundai Motor Group to Pioneer Hong Kong's Hydrogen Economy, Accelerating Asia-Pacific Expansion
[Picked up by 290 media titles]
2026-05-18 15:30Autonomous AI marketing platform Protaigé launches with Maia, the world's first AI Account Director that operates within the flow of work
[Picked up by 281 media titles]
2026-05-20 09:15