Medical/Pharmaceuticals
Harbour BioMed Announces 2024 Interim Results
CAMBRIDGE, Mass., ROTTERDAM, Netherlands and SUZHOU, China, Aug. 28, 2024 /PRNewswire/ -- Harbour BioMed ("HBM", or the "Company"; HKEX: 02142), a global biopharmaceutical company committed to the discovery, development, and commercialization of novel antibody therapeutics focusing on oncology an...
Hyundai Bioscience to conduct phase 3 clinical trial for high-risk group of COVID-19 patients with the goal of emergency use authorization
* "Xafty® can be the only oral COVID-19 treatment that can be prescribed to high-risk group patients who cannot take Paxlovid®" * "While maintaining the current process for emergency use authorization for mild-to-moderate COVID-19 patients, Hyundai Bioscience will expedite Phase 3 clinical tr...
Achieva Medical Entered into an Exclusive Distribution Agreement with NowYon Medical
SUZHOU, China, Aug. 28, 2024 /PRNewswire/ -- Peijia Medical (HKG:9996), a leading Chinese domestic player in the high-growth transcatheter valve therapeutics and neurovascular interventions markets, announced that its wholly-owned subsidiary, Achieva Medical Limited ("Achieva Medical"), entered ...
Yunovia Announces IND Approval by the MFDS to Initiate Phase 1 MAD Study for the Small Molecule GLP-1 Agonist
SEOUL, South Korea, Aug. 27, 2024 /PRNewswire/ -- Yunovia, a drug R&D subsidiary of Ildong Pharmaceutical Group, announced on the 26th that the Ministry of Food and Drug Safety (MFDS) of Korea has cleared the IND application of Phase 1 Multiple Ascending Dose (MAD) study for ID110521156, an oral...
Shilpa Medicare Makes History: First Indian Company Completes Phase 1 Trial for Recombinant Human Albumin (rHA) 20%
RAICHUR, India, Aug. 28, 2024 /PRNewswire/ -- In a major breakthrough, Shilpa
Medicare Limited
Jenscare, with innovative TTVR, releases 2024H1 interim results
BEIJING, Aug. 28, 2024 /PRNewswire/ -- Jenscare Scientific Co., Ltd. ("Jenscare" or the "Company") (HKEX: 9877), an innovative medical device company dedicated to interventional treatment for structural heart diseases with TTVR breakthroughs, released interim results for 2024H1 endedJune 30, 2024...
Everest Medicines Announces Interim Results for First Half of 2024
SHANGHAI, Aug. 28, 2024 /PRNewswire/ -- Everest Medicines (HKEX 1952.HK, "Everest", or the "Company"), a biopharmaceutical company focused on the development, manufacturing and commercialization of innovative medicines and vaccines, today announced its interim results for the first half of 2024 a...
Telix Submits NDA for TLX101-CDx (Pixclara®) Brain Cancer Imaging Agent
MELBOURNE, Australia, Aug. 28, 2024 /PRNewswire/ -- Telix Pharmaceuticals Limited (ASX: TLX, Telix, the Company) today announces it has submitted a New Drug Application (NDA) tothe United States (U.S.) Food and Drug Administration (FDA) for TLX101-CDx, (Pixclara®[1], 18F-floretyrosine or 18F-FET)...
Unique International Collaboration Brings Breakthrough Oral Cholera Vaccine to Market
Singapore-based Hilleman Laboratories, along with Bharat Biotech, confirm launch of new cholera vaccine, HILLCHOL® Success provides "template for the internationalization of future vaccine and biologics development fromSingapore" – Dr. Raman Rao, CEO, Hilleman Laboratories Hilleman Laboratories ...
Boan Biotech Announces 2024 Half Year Results: Revenue Grows by 39%, Net Profit Increases by RMB 180 Million
YANTAI, China, Aug. 27, 2024 /PRNewswire/ -- Boan Biotech (6955.HK) today announced its 2024 half-year results and latest developments. During the reporting period, the company's total revenue was RMB 363 million, up 39% year-over-year. In this, the revenue from product sales wasRMB 332 million,...
GenScript Subsidiary Legend Biotech Achieves Breakthrough with Cilta-cel Approval in China, Offering New Hope for Multiple Myeloma Patients
PISCATAWAY, N.J., Aug. 27, 2024 /PRNewswire/ -- Legend Biotech (NASDAQ: LEGN), a subsidiary of GenScript Biotech Corporation (hereinafter referred to as "GenScript"), a global leader in life sciences research and manufacturing services, today announced that they have received approval from the Ch...
Keymed Biosciences Announces Interim Results for First Half of 2024
CHENGDU, China , Aug. 27, 2024 /PRNewswire/ -- Keymed Biosciences Inc. (HKEX: 02162) today announced its interim results for the first half of 2024, along with a corporate update. Rapid development of our pipeline products Stapokibart (CM310) (IL-4Rα antibody) * In June 2024, the long-term e...
Sleep Specialists and Industry Leaders Converge at HoneyNaps Symposium
* The 1st HoneyNaps Sleep & Tech Symposium * Presentation of the 'SLEEP 2024 White Paper' and the Latest Advances in Sleep Research & Technology * Unveiling of HoneyNaps' New Brand Identity BOSTON, Aug. 27, 2024 /PRNewswire/ -- HoneyNapsUSA, Inc., an AI-driven sleep data analysis company, p...
Currax Pharmaceuticals: CONTRAVE®/MYSIMBA® Demonstrates Positive Cardiovascular Safety in a Large, Real-World Evidence Study
BRENTWOOD, Tenn., Aug. 27, 2024 /PRNewswire/ -- Currax Pharmaceuticals LLC ("Currax") today announced the results of a Cardiovascular Health Outcomes Analysis (HOA). The results showed there is no evidence of excess cardiovascular risk and no statistically significant difference in major adverse...
111, Inc. to Participate in Fireside Chat with Water Tower Research on September 5, 2024
SHANGHAI, Aug. 27, 2024 /PRNewswire/ -- 111, Inc. ("111" or the "Company") (NASDAQ: YI), a leading tech-enabled healthcare platform company committed to reshaping the value chain of healthcare industry by digitally empowering the upstream and downstream inChina, today announced that it will parti...
JW Therapeutics Announces NMPA Approval of the Supplemental Biological License Application for Carteyva® in Adult Patients with Relapsed or Refractory Mantle Cell Lymphoma
SHANGHAI, Aug. 27, 2024 /PRNewswire/ -- JW Therapeutics (HKEx: 2126), an independent and innovative biotechnology company focusing on developing, manufacturing and commercializing cell immunotherapy products, announced that the National Medical Products Administration (NMPA) ofChina has approv...
HKBU research identifies potential of phosphocholine in counteracting PM2.5 toxicity in lung diseases
HONG KONG, Aug. 27, 2024 /PRNewswire/ -- A research team led by Hong Kong Baptist University (HKBU) has discovered that phosphocholine, an important component in the body's metabolism, could counteract the adverse effects of PM2.5 toxicity on lung cells. This discovery highlights the therapeutic ...
ArkBio Presented at the 8th Annual IPF Summit and Announced Completion of Patient Enrollment for Phase 2 Clinical Trial of AK3280, a Next-Generation Drug for IPF Treatment
BEIJING, Aug. 27, 2024 /PRNewswire/ -- Shanghai Ark Biopharmaceutical Co., Ltd. ("ArkBio"), a privately held, clinical-stage biopharmaceutical company today announced that Dr.Jim Wu, Chief Executive Officer of ArkBio, gave an oral presentation entitled"Development of Next Generation Broadly Activ...
Fujitsu tackles 'drug loss' in Japan through ecosystem to accelerate digitalization of clinical trials
Fujitsu forms strategic partnership with Paradigm and will provide offerings that leverage the power of AI TOKYO, Aug. 27, 2024 /PRNewswire/ -- Fujitsu today announced that it will begin initiatives to attract global clinical trials toJapan and tackle the 'drug loss' issue by working with pharma...
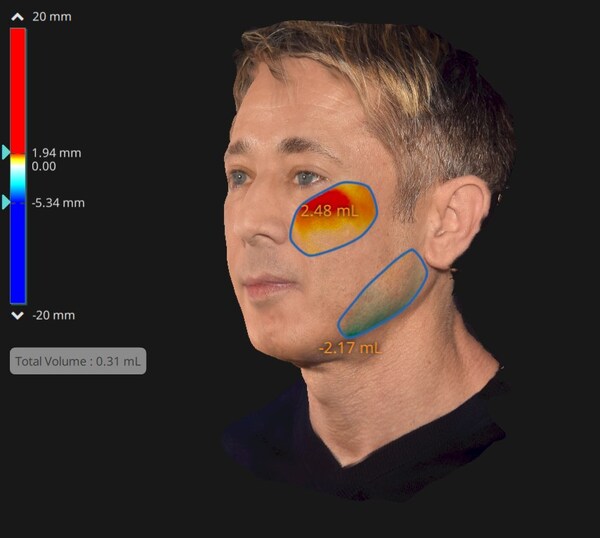
Experts Agree On the Longevity of EMFACE's Non-Invasive Face-Lifting Effects
PRAGUE, Aug. 27, 2024 /PRNewswire/ -- Continuous research by Dr. Yael Halaas (
USA) and Dr. Woraphong Manuskiatti (Thailand) has unveiled the first evidence
on the longer-term effectiveness of EMFACE
Week's Top Stories
Most Reposted
Marina Bay precinct partners UOB, Marina Bay Sands and Singapore Tourism Board, together with Disney Cruise Line, to illuminate Singapore's skyline with a fireworks sky show
[Picked up by 328 media titles]
2026-02-19 14:30NextFin Asia: A New Dedicated Fund for the Catapult: Inclusion SE Asia Program to Further Scale Inclusive Finance Fintechs in ASEAN
[Picked up by 310 media titles]
2026-02-23 08:00Vitafoods Asia 2026 Expands by 30%: A Bigger, More Dynamic Trade Event with Exciting New Features & Increased International Participation
[Picked up by 289 media titles]
2026-02-23 10:09Little Artists Art Studio, Singapore Shines at Art Capital 2026
[Picked up by 280 media titles]
2026-02-17 19:12Kung Fu Meets Spring -- Unitree Spring Festival Gala Robots Present "Cyber Real Kung Fu" in the Year of the Horse
[Picked up by 259 media titles]
2026-02-17 14:16