Pharmaceuticals
ReviR Therapeutics Announces First Participant Dosed in Phase 1 Study and Secures CDE Clearance for Two Orphan Indications in China, Supporting Future U.S. Expansion Strategy
Milestone marks the first clinical program for Charcot-Marie-Tooth disease and Vanishing White Matter disease. SAN FRANCISCO and SHANGHAI, March 2, 2026 /PRNewswire/ -- ReviR Therapeutics, a clinical-stage biotechnology company pioneering AI-driven RNA-modulating small molecules to treat rare an...
Halia Therapeutics Announces Presentation at the BIO Investment & Growth Summit
LEHI, Utah, March 2, 2026 /PRNewswire/ -- Halia Therapeutics, a clinical-stage biopharmaceutical company advancing therapies grounded in human biology, today announced that it will deliver a company presentation at the BIO Investment & Growth Summit. David Bearss, Ph.D., Chief Executive Officer ...
The largest pharma show in Asia makes its return to Shanghai this June 2026
SHANGHAI, March 2, 2026 /PRNewswire/ -- CPHI & PMEC China 2026 is set to take
place from16 to 18 June 2026 at the Shanghai New International Expo Centre
(SNIEC), building on its extremely successful run over the last few years.
XPOVIO® Receives Reimbursement Approval in South Korea for a Second Multiple Myeloma Indication
- XPOVIO® is the first XPO1 inhibitor approved for reimbursement by South Korea's National Health Insurance Service (NHIS) for the treatment of adult patients with multiple myeloma (MM). - In South Korea, XPOVIO® has been approved for three indications across MM and diffuse large B-cell lymphoma...
Jaypirca® (Pirtobrutinib) Approved in China for the Treatment of Relapsed or Refractory Chronic Lymphocytic Leukemia or Small Lymphocytic Lymphoma
SAN FRANCISCO and SUZHOU, China, March 1, 2026 /PRNewswire/ -- Innovent Biologics, Inc. ("Innovent") (HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures, and commercializes high-quality medicines for the treatment of oncologic, autoimmune, cardiovascular and metabol...
Lynk Pharmaceuticals Announces Positive Phase III Topline Data of Zemprocitinib in Moderate-to-Severe Atopic Dermatitis
SHANGHAI, HANGZHOU, China and BOSTON, March 2, 2026 /PRNewswire/ -- Lynk Pharmaceuticals Co., Ltd. ("Lynk Pharmaceuticals"), a clinical-stage innovative drug development company focused on therapies for immune and inflammatory diseases, today announced positive topline results from its Phase III ...
Investor Education Webinar
The Future of Prostate Imaging: What do physicians want? MELBOURNE, Australia and INDIANAPOLIS, March 2, 2026 /PRNewswire/ -- Telix Pharmaceuticals Limited (ASX: TLX, NASDAQ: TLX, "Telix") invites investors to join a webinar with key opinion leaders to discuss a physician's perspective on innova...
Earendil Labs Enters Strategic Collaboration with WuXi XDC on WuXiTecan-2 Payload-Linker Technology Platform
WILMINGTON, Del., Feb. 27, 2026 /PRNewswire/ -- Earendil Labs, a global leader in AI-driven research and development of next-generation biologics therapeutics, today announced a strategic collaboration with WuXi XDC Cayman Inc. ("WuXi XDC", stock code: 2268.HK) on WuXi XDC's proprietary WuXiTecan...
ImmVira's Oncolytic Product MVR-T3011 Expanded to BCG-Naïve Bladder Cancer Patients for the First Time with Clinical Data Presented at the 2026 ASCO GU Conference
SUZHOU, China, Feb. 26, 2026 /PRNewswire/ -- Intravesical BCG is the standard of care (SOC) for BCG-naïve high-risk non-muscle-invasive bladder cancer (NMIBC) patients. However, the scarcity of BCG products has become a global phenomenon, and coupled with the side effects of BCG therapy itself,me...
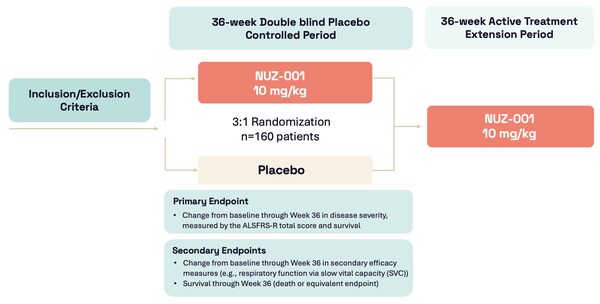
Neurizon Initiates Dosing of NUZ-001 in HEALEY ALS Platform Trial
Highlights: * First participant enrolled and dosed with NUZ-001 in Regimen I of the HEALEY ALS Platform Trial evaluating NUZ-001 for the treatment of ALS * Approximately 160 participants with ALS will be enrolled in a 36-week randomised,double-blind, placebo-controlled adaptive Phase 2/3 clin...
ArisGlobal Launches NavaX Translation to Eliminate Manual Translation in Global Pharmacovigilance
Strategic partnership with TransPerfect Life Sciences embeds certified,
pharma-grade translation directly into LifeSphere Safety workflows
BOSTON, Feb. 26, 2026 /PRNewswire/ -- ArisGlobal
SK bioscience, IDT Biologika and Vaxxas Selected for Next-Generation Influenza Vaccine development Financed by the European Union
* SK–IDT–Vaxxas consortium establishes strategic entry point for high-density microarray patch (HD-MAP) vaccines inEurope to improve vaccine performance and access. * Parallel development of high-immunogenicity HD-MAP vaccines for seasonal and pandemic influenza threats. INCHEON, South Korea,...
Illumina partners go beyond the genome, driving cancer breakthroughs using spatial transcriptomics, epigenomics, and proteomics
At AGBT, researchers reveal new findings from studies leveraging Illumina's multiomics solutions SAN DIEGO, Feb. 26, 2026 /PRNewswire/ -- Illumina, Inc. (NASDAQ: ILMN) announced new customer breakthroughs in oncology powered by Illumina's spatial transcriptomics, 5-base sequencing, and proteomi...
Sanyou Biopharmaceuticals Announces New Strategic Investor; Liferiver Biotech Increases Its Stake, and Gree Group's Yanghe Investment Joins as a Strategic Shareholder
SHANGHAI, Feb. 24, 2026 /PRNewswire/ -- Sanyou Biopharmaceuticals (Shanghai) Co., Ltd. ("Sanyou Bio"), a biotech company powered by super-trillion-molecule libraries and AI-driven technologies, today announced the completion of a significant equity restructuring. The shares previously held by Hai...
Sciwind Biosciences Partners with Pfizer China to Commercialize its Biased GLP-1 in China
HANGZHOU, China, Feb. 24, 2026 /PRNewswire/ -- Hangzhou Sciwind Biosciences Co., Ltd. ("Sciwind Biosciences") today announced a strategic commercialization collaboration with Pfizer China for Ecnoglutide injection (Ecnoglutide), a new–generation cAMP–biased GLP–1 receptor agonist. Under the agree...
iRegene Hits Dual Global Firsts: U.S. Patient Dosed in Phase IIa for NouvNeu001 in Parkinson's and First Enrollment in China's Randomized MSA Trial for NouvNeu004
CHENGDU, China, Feb. 23, 2026 /PRNewswire/ -- The global race to treat neurodegenerative diseases has reached a new tempo as iRegene Therapeutics pushes its cell therapy pipeline into key clinical stages in the U.S. andChina. Concurrently, the company's "AI + Chemical Induction" platform saw its...
Formosa Pharmaceuticals Announces Licensing Agreement with Arrotex, for Commercialization of Clobetasol Propionate Ophthalmic Suspension for the Treatment of Inflammation and Pain Following Ocular Surgery
TAIPEI, Feb. 23, 2026 /PRNewswire/ -- Taiwan-based Formosa Pharmaceuticals ("Formosa", 6838.TW) announced today that the company has entered into an exclusive licensing agreement with Arrotex Pharmaceuticals Pty Ltd. ("Arrotex"), for exclusive rights to the commercialization of clobetasol propio...
CStone Announces MHRA Approval in UK for Sugemalimab in Stage III NSCLC
* Following approval by the European Commission (EC), sugemalimab has received a new indication approval from the UK Medicines and Healthcare products Regulatory Agency (MHRA) for stage III non-small cell lung cancer (NSCLC). This marks the second indication approved for the product in the UK. ...
FY 2025 Results: Strong Commercial Growth, Focused Pipeline Investment
MELBOURNE, Australia and INDIANAPOLIS, Feb. 20, 2026 /PRNewswire/ -- Telix Pharmaceuticals Limited (ASX: TLX, NASDAQ: TLX, "Telix") today announces its financial results for the year ended December 31, 2025. FY 2025 key results1 Group performance2: Double-digit revenue growth and positive adju...
Cambrex Announces Cornell Professor Song Lin as Recipient of the 2025 Snapdragon Prize for Innovation in Chemistry Technology
EAST RUTHERFORD, N.J., Feb. 19, 2026 /PRNewswire/ -- Cambrex, a leading global contract development and manufacturing organization (CDMO) and Snapdragon Chemistry, a Cambrex company, are proud to announce that Dr. Song Lin, Tisch University Professor in chemistry and chemical biology at Cornell U...
Week's Top Stories
Most Reposted
Visa and Trip.com Group Ink Strategic Collaboration, Enabling Consumers to Chase Their Passions Through Travel
[Picked up by 308 media titles]
2026-05-19 18:10Two-day Global Prosperity Summit 2026 officially opens
[Picked up by 308 media titles]
2026-05-19 23:59Hong Leong Bank Awarded Best Managed Bank in Malaysia by The Asian Banker, Group MD/CEO Kevin Lam Named Best Bank CEO in Malaysia
[Picked up by 293 media titles]
2026-05-15 17:00Hyundai Motor Group to Pioneer Hong Kong's Hydrogen Economy, Accelerating Asia-Pacific Expansion
[Picked up by 290 media titles]
2026-05-18 15:30Two‑day Global Prosperity Summit 2026 concludes successfully
[Picked up by 285 media titles]
2026-05-21 22:13