Health Care/Hospital
YishengBio Announces PIKA recombinant COVID-19 vaccine received Phase I clinical trial IND clearance from New Zealand
BEIJING, Aug. 9, 2021 /PRNewswire/ -- YishengBio Co., Ltd ("YishengBio") today announced that it received IND clearance for PIKA recombinant COVID-19 vaccine (CHO cell, S Protein) to conduct Phase I clinical trial from New Zealand Medicines and Medical Devices Safety Authority ("MEDSAFE"), which ...
Life Sciences' Contribution to the US Digital Therapeutics Market to Grow the Fastest amongst all End Users
The US digital therapeutics market is estimated to witness more than three-and-a-half-fold growth reaching$4.54 billion by 2025, at a CAGR of 29.8%, finds Frost & Sullivan SANTA CLARA, Calif., Aug. 9, 2021 /PRNewswire/ -- Frost & Sullivan's recent analysis finds that digital therapeutics (DTx) i...
Thailand's Jan-June Investment Applications Reach USD12 billion, as FDI Quadruple, E&E and Medical Sectors Continue to Expand, BOI says
BANGKOK, Aug. 9, 2021 /PRNewswire/ -- In the first six months of 2021, Thailand's investment applications increased 14% from the year earlier period in terms of the number of projects, and 158% in combined value, led by increasing foreign direct investment (FDI) applications, sustained growth in ...
Antengene and MindRank AI Enter into Collaboration to Advance the Development of Difficult-to-Drug Molecular Targets
SHANGHAI and HONG KONG, Aug. 9, 2021 /PRNewswire/ -- Antengene Corporation Limited ("Antengene", SEHK: 6996.HK), a leading innovative biopharmaceutical company dedicated to discovering, developing and commercializing global first-in-class and/or best-in-class therapeutics in hematology and oncolo...
China NMPA Approves Phase II Clinical Trial of ASC40 for the Treatment of Patients with Acne
HANGZHOU and SHAOXING, China, Aug. 9, 2021 /PRNewswire/ -- Ascletis Pharma Inc. (HKEX code: 1672) today announces that China National Medical Products Administration (NMPA) has approved the Phase II clinical trial application of ASC40 for the treatment of patients with moderate to severe acne. T...
Innovent Announces the Establishment of the Scientific Advisory Board
SAN FRANCISCO and SUZHOU, China, Aug. 9, 2021 /PRNewswire/ -- Innovent Biologics, Inc. ("Innovent") (HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures and commercializes high-quality medicines for the treatment of cancer, metabolic, autoimmune and other major dise...
Endurance Longevity Announces Positive Profit Alert for Interim Results 2021 and Other Exciting Business Updates
HONG KONG, Aug. 6, 2021 /PRNewswire/ -- Endurance RP Limited's ("Endurance Longevity" or the "Company" and together with its subsidiaries, the "Group"; stock code: 0575.HK) is pleased to announce a positive profit alert for the interim results 2021 for the six month period ended30 June 2021. The ...
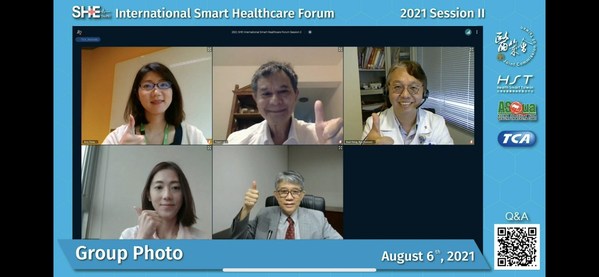
AI and Big Data Join Hands to Assist Physicians in Diagnosis and Reporting
TAIPEI, Aug. 6, 2021 /PRNewswire/ -- With the advent of the 5G era and the growing popularity of big data and artificial intelligence applications, the Joint Commission ofTaiwan (JCT) is hosting Smart Healthcare Expo Taipei 2021: International Smart Healthcare Forum (Session 2) with a focus on tw...
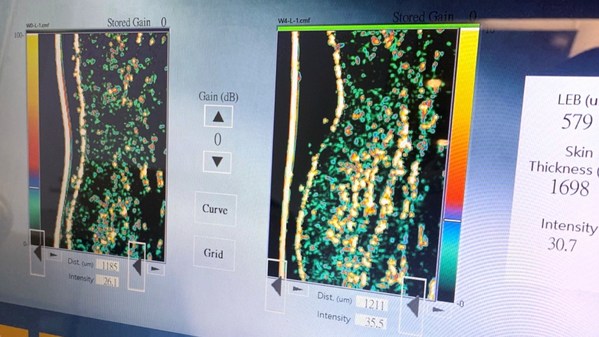
Taiwan's medical device company challenges the limit of science, launching the most expensive collagen in the contemporary market
TAIPEI, Aug. 6, 2021 /PRNewswire/ -- As consumers around the world increase their awareness of using beauty and health products, collagen products have become the most popular item. Various manufacturers continue to innovate in accordance with consumer demand. According to Research and Markets, w...
ORI Capital's Portfolio Company Pillar Receives FDA Premarket Approval for its oncoReveal(TM) Dx Lung and Colon Cancer Test
HONG KONG, Aug. 6, 2021 /PRNewswire/ -- ORI Capital, a quantamental healthcare fund manager focused on investing in disruptive healthcare companies globally, announced today the U.S. Food and Drug Administration (FDA) has given its portfolio company, Pillar Biosciences Inc. ("Pillar"), Premarket ...
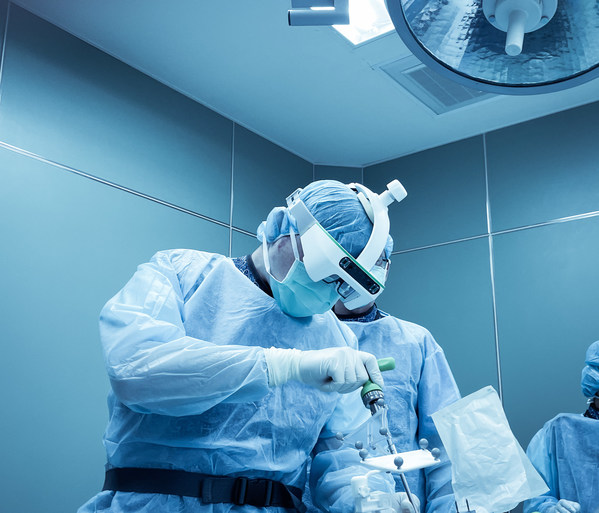
SURGLASSES' SPINE NAVIGATION SYSTEM CADUCEUS S - An innovation greatly influencing minimally invasive surgeries
TAICHUNG, Aug. 6, 2021 /PRNewswire/ -- Taiwan Main Orthopaedic Biotechnology Co. Ltd (SURGLASSES),Taiwan, a company striving to bring a difference into the real time surgical procedure after the introduction of the first AR assisted Head Mounted Display Foresee-X, is now coming up with a new prod...
Updated Overall Survival Data and Biomarker Results from Sintilimab ORIENT-11 Study in First-Line Nonsquamous Non-Small Cell Lung Cancer Published in the Journal of Thoracic Oncology
SAN FRANCISCO and SUZHOU, China, Aug. 6, 2021 /PRNewswire/ -- Innovent Biologics, Inc. ("Innovent") (HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures and commercializes high-quality medicines for the treatment of oncology, metabolic, autoimmune and other major dis...
Novavax Announces COVID-19 Vaccine Booster Data Demonstrating Four-Fold Increase in Neutralizing Antibody Levels Versus Peak Responses After Primary Vaccination
* Single booster dose at 6 months of NVX-CoV2373 increased wild-type neutralizing antibodies more than 4-fold versus primary vaccination series * Six-fold increase in cross-reactive functional antibodies to Delta variant compared to primary vaccination series * Analysis of sera from primary ...
Novavax and Serum Institute of India Announce Submission to Regulatory Agencies in India, Indonesia, Philippines for Emergency Use Authorization of Novavax' Recombinant Nanoparticle COVID-19 Vaccine
- Novavax and Serum Institute of India file regulatory submissions to the Drugs Controller General ofIndia and to regulatory agencies in Indonesia and Philippines for Novavax' recombinant nanoparticle COVID-19 vaccine - Filing for World Health Organization Emergency Use Listing expected in Augus...
Advanced Instruments to Acquire Solentim, a Trusted Global Leader in Cell Imaging for Cell Line Development and Clone Characterization
NORWOOD, Mass., Aug. 6, 2021 /PRNewswire/ -- Advanced Instruments, a leading manufacturer of analytical instruments and services for bioprocessing, today announced the execution of a definitive agreement to acquire Solentim, a trusted global leader of solutions for the isolation and characterizat...
Brii Biosciences Announces the Completion of Enrollment in the Phase 3 National Institutes of Health (NIH) ACTIV-2 Trial, Evaluating the BRII-196 and BRII-198 Monoclonal Neutralizing Antibody Combination for the Treatment of COVID-19
846 outpatients at high risk of clinical progression have been enrolled in the ACTIV-2 phase 2/3 clinical study, from sites inthe United States, Brazil, South Africa, Mexico and Argentina. The participants are being evaluated for the combined endpoint of hospitalizations and death relative ...
ArisGlobal Lauded by Frost & Sullivan for Enabling Life Sciences Companies to Accelerate R&D with Its LifeSphere® Platform
LifeSphere® ably supports clients by expediting decision-making, maintaining compliance, and streamlining collaborations among cross-functional teams SANTA CLARA, Calif., Aug. 5, 2021 /PRNewswire/ -- Based on its recent analysis of the North American clinical development and pharmacovigilance so...
Ortho Clinical Diagnostics Expands COVID-19 Test Offerings with Emergency Use Authorization (EUA) of Total Nucleocapsid Antibody Test
-- Ortho's new VITROS® Anti-SARS-CoV-2 Total N test detects the individual's immune response to the COVID-19 virus and is intended to help clinicians understand if their patient had a recent or prior COVID-19 infection. -- Ortho is the first company to have an EUA for both a quantitative IgG a...
AXA launches first-in-market "SurgiCare Surgical Insurance Plan" to provide lump-sum payout for VHIS defined surgeries
HONG KONG, Aug. 5, 2021 /PRNewswire/ -- AXA Hong Kong (AXA) launches "SurgiCare Surgical Insurance Plan" ("SurgiCare"). "SurgiCare" is the first-in-market surgical insurance plan based on the four levels under the existing Voluntary Health Insurance Scheme (VHIS) schedule of surgical procedures –...
Insilico Medicine and Usynova Announce Strategic Partnership on Accelerating R&D of Small Molecule Innovative Drugs with AI
SHANGHAI, Aug. 5, 2021 /PRNewswire/ -- Insilico Medicine, an industry leader in
end-to-end artificial intelligence for target discovery, small molecule
chemistry, and clinical development, and Usynova announced to reach strategic
cooperation in advancing the development of novel therapies.
Week's Top Stories
Most Reposted
Protiviti Transforms Cyber Risk Consulting with CYFIRMA's Advanced Intelligence-Led Cybersecurity
[Picked up by 324 media titles]
2024-04-15 09:00Sephora appoints Xia Ding as Managing Director of Sephora Greater China
[Picked up by 309 media titles]
2024-04-15 17:33AI-DOL: Revolutionising the Future of Entertainment with AI-Powered Virtual Idols
[Picked up by 295 media titles]
2024-04-12 15:14Animated Life in Yunnan
[Picked up by 275 media titles]
2024-04-18 14:00Allianz Partners Introduces Instant Online Chat for HCF Travel Insurance Customers
[Picked up by 275 media titles]
2024-04-15 08:00