Health
APL-2301, a compound developed by Asieris for the treatment of Acinetobacter baumannii infection, was approved for Phase I clinical trials in Australia
SHANGHAI, Dec. 24, 2023 /PRNewswire/ -- Asieris Pharmaceuticals (Stock Code: 688176.SH), a global biopharma company specializing in discovering, developing and commercializing innovative drugs for the treatment of genitourinary tumors and other related diseases, announced that its product, APL-23...
GenSci Launches Global Innovation Hub in Shanghai
The Establishment of the Facility Demonstrates the Company's Commitment to Delivering Differentiated Solutions to Gynecology and Pediatrics Challenges SHANGHAI, Dec. 22, 2023 /PRNewswire/ -- Changchun GeneScience Pharmaceutical ("GenSci"), a subsidiary of Changchun High-Tech Industries (Group), h...
Yoshitsu Co., Ltd Reports First Six Months of Fiscal Year 2024 Financial Results
TOKYO, Dec. 22, 2023 /PRNewswire/ -- Yoshitsu Co., Ltd ("Yoshitsu" or the "Company") (Nasdaq: TKLF), a retailer and wholesaler of Japanese beauty and health products, sundry products, luxury products, electronic products, as well as other products inHong Kong, mainland China, Japan, North America...
TransThera Announces the Global Multicenter Phase 3 Clinical Trial Completed First Patient Dosing in the US Evaluating Tinengotinib in FGFRi Relapsed/Refractory Patients with Cholangiocarcinoma
NANJING, China and GAITHURSBURG, Md., Dec. 21, 2023 /PRNewswire/ -- TransThera, a clinical-stage biopharmaceutical company dedicated to innovating differentiated drugs globally, today announces the first patient has been dosed in the US for the Phase 3 trial FIRST-308 of tinengotinib (TT-00420), ...
BioCity announces the first patient dosed with its anti-TIM-3 mAb BC3402 in Combination with IMFINZI for the Treatment of Advanced Hepatocellular Carcinoma in a Phase Ib/II Trial
WUXI, China, Dec. 20, 2023 /PRNewswire/ -- BioCity Biopharma today announced dosing of the first patient in a Phase Ib/II clinical trial of its anti-TIM-3 monoclonal antibody (mAb) BC3402 in combination with IMFINZI (durvalumab) for the treatment of advanced hepatocellular carcinoma (HCC) inCh...
YS Biopharma Responds to Unauthorized Press Release Regarding Extraordinary General Meeting
GAITHERSBURG, Md., Dec. 20, 2023 /PRNewswire/ -- YS Biopharma Co., Ltd. (NASDAQ: YS) ("YS Biopharma" or the "Company"), a global biopharmaceutical company dedicated to discovering, developing, manufacturing, and delivering new generations of vaccines and therapeutic biologics for infectious disea...
/DISREGARD RELEASE: YS Biopharma/
We are advised by YS Biopharma Co., Ltd. (NASDAQ:YS) that journalists and other readers should disregard the news release, "YS Biopharma Co., Ltd. to Hold Extraordinary General Meeting onDecember 28, 2023", issued on December 20, 2023 by YS Biopharma over PR Newswire, as the release contained err...
CellOrigin announced treatment of the first patient with CAR-M in China and reported the second generation of CAR-M for solid tumors
HANGZHOU, China, Dec. 20, 2023 /PRNewswire/ -- Recently, a CAR-macrophage therapy product (CAR-M) SY001 from CellOrigin Biotechnology (Hangzhou) Co., Ltd. was dosed to the first patient in a hospital inChina. "A single center, single-arm, dose-escalation, exploratory clinical trial to examine t...
Genevoyager Unveils Gene Therapy CDMO Facility in Milestone Move
PHILADELPHIA, Dec. 20, 2023 /PRNewswire/ -- Genevoyager (Wuhan) Co., Ltd. (Genevoyager), a leading provider of one-stop CRO/CDMO services for gene therapy products, proudly announces the official opening of its Contract Development and Manufacturing Organization (CDMO) facility. This significant ...
HanAll Biopharma, Daewoong Pharmaceutical and NurrOn Pharmaceuticals to Showcase Ongoing Parkinson's Disease Program at the 7th Annual Sachs Associates Neuroscience Innovation Forum
ROCKVILLE, Md., Dec. 20, 2023 /PRNewswire/ -- HanAll Biopharma (KRX: 009420.KS), Daewoong Pharmaceutical (KRX: 069620.KS), and NurrOn Pharmaceuticals announced their participation in the 7th Annual Sachs Associates Neuroscience Innovation Forum. The event is scheduled to take place onJanuary 7th...
InxMed Enters License Agreement with Escugen to Develop Next-Generation ADCs
NANJING, China, Dec. 19, 2023 /PRNewswire/ -- InxMed, a clinical-stage biotechnology company dedicates on developing innovative therapies against drug resistance, and Escugen, a clinical-stage antibody–drug conjugate (ADC) company, today announced that InxMed licensed EZWi-Fit® linker-payload pl...
Standigm named as a Tech Innovator in Generative AI in Drug Discovery use case
SEOUL, South Korea, Dec. 19, 2023 /PRNewswire/ -- Standigm
New Drug Application of Mabwell's 8MW0511 for Injection Accepted by NMPA
SHANGHAI, Dec. 19, 2023 /PRNewswire/ -- Mabwell (688062.SH), an innovative biopharmaceutical company with entire industry chain, announced that the new drug application of its recombinant (yeast-secreted) human serum albumin-human granulocyte colony-stimulating factor (I) fusion protein for injec...
Tianjin's Youthful Energy Sparks Citywide Revival
TIANJIN, China, Dec. 19, 2023 /PRNewswire/ -- A report from China Daily: Tianjin, a city in North China, is undergoing a dynamic transformation driven by its vibrant young people. China Daily Potside Chats hosts, Nathan and Stephanie, recently explored the city's cutting-edge developments led by ...
Tsingke Congratulates iGEM 2023 SYSU-Software on Winning GOLD MEDAL for BioRocket Project
BEIJING and PARIS, Dec. 18, 2023 /PRNewswire/ -- The 2023 International Genetically Engineered Machine Competition (iGEM), the global synthetic biology competition, concluded onNovember 5, 2023. Over 7,000 students from high schools and universities, organized into more than 400 teams worldwide, ...
Biorithm Secures $3.5 Million in Series A Funding to Advance Maternal Care Worldwide
New funds to fortify US market entry and growth strategy, as well as advance breakthrough research CAMBRIDGE, Mass., Dec. 18, 2023 /PRNewswire/ -- Biorithm, a global women's health medtech, has closed$3.5 million in Series A funding, co-led by Adaptive Capital Partners and SEEDS Capital. With a ...
Neuren Phase 2 trial shows significant improvements in Phelan-McDermid syndrome
Highlights: * Significant improvement was assessed by both clinicians and caregivers across multiple efficacy measures * Improvements were consistently seen across clinically important aspects of Phelan-McDermid syndrome, including communication, behaviour, cognition/learning and socialis...
NUS researchers develop an innovative and flexible method to study immune cell capabilities
This simple and efficient method can directly identify and sort immune cells involved in cell-mediated cytotoxicity SINGAPORE, Dec. 17, 2023 /PRNewswire/ -- Our body's immune system is a complex network of organs, cells, and proteins that work in synchrony to protect our bodies against infection...
GC Biopharma Announces US FDA Approval for ALYGLO™ (Immune Globulin Intravenous, Human-stwk) 10% Liquid for Adults with Primary Humoral Immunodeficiency (PI)
YOGIN, South Korea, Dec. 17, 2023 /PRNewswire/ -- GC Biopharma Corp (006280.KS) today announces that the US Food and Drug Administration (FDA) has approved ALYGLO (immune globulin intravenous, human-stwk) 10% Liquid, formerly referred to as "GC5107," for the treatment of adult patients aged 17...
GeneQuantum and BioMap establish a strategic collaboration to jointly develop a series of next-generation ADC therapeutics
SUZHOU, China, Dec. 17, 2023 /PRNewswire/ -- Recently, GeneQuantum Healthcare (GQ), a global leader in ADC new drug development focused on innovative bioconjugation technologies, and BioMap, a global leader in life science AI Foundation protein-centric Large Language Models, announced that the...
Week's Top Stories
Most Reposted
Mastercard goes OTP-free in APAC for faster, safer online transactions
[Picked up by 326 media titles]
2024-11-06 09:00Brankas Launches Integrated APAC Open Banking Compliance Solution with ADVANCE.AI's eKYC Solution
[Picked up by 319 media titles]
2024-11-07 09:00Going Global: DCITS Embarks on International Expansion at Singapore Fintech Festival
[Picked up by 308 media titles]
2024-11-12 09:00Ubiqconn Technology to Showcase Latest Marine Solutions at the 2024 International WorkBoat Show in New Orleans
[Picked up by 288 media titles]
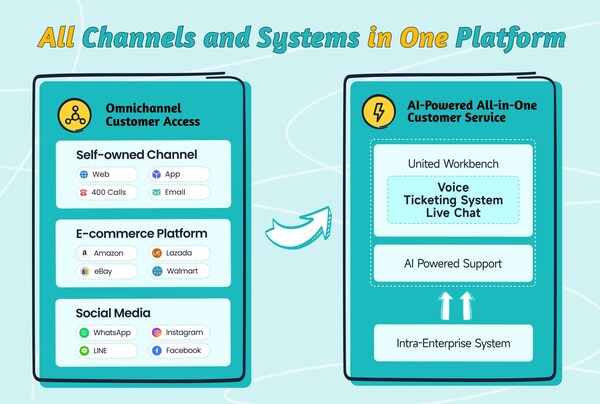
2024-11-11 21:00Sobot Introduces its All-in-One Solution at GITEX Global 2024
[Picked up by 284 media titles]
2024-11-12 11:00