Neurizon Initiates Dosing of NUZ-001 in HEALEY ALS Platform Trial
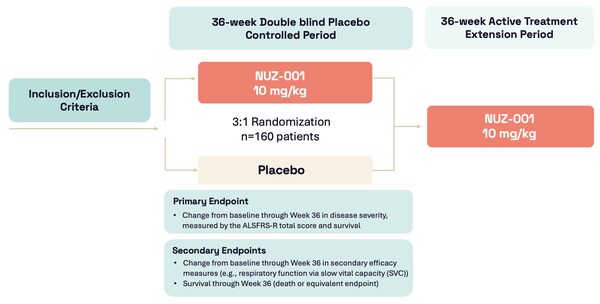
Highlights: * First participant enrolled and dosed with NUZ-001 in Regimen I of the HEALEY ALS Platform Trial evaluating NUZ-001 for the treatment of ALS * Approximately 160 participants with ALS will be enrolled in a 36-week randomised,double-blind, placebo-controlled adaptive Phase 2/3 clin...
FDA Clears NUZ-001 for Entry into HEALEY ALS Platform Trial
Highlights: * FDA has completed their review of the Neurizon NUZ-001 regimen to the HEALEY ALS Platform Trial Master Protocol * This marks the official entry of NUZ-001 as Regimen I in the HEALEY ALS Platform Trial * Next steps include obtaining single IRB approval, site initiations and cl...
FDA Lifts Clinical Hold on NUZ-001
Highlights: * U.S. FDA lifts the Clinical Hold on NUZ‑001, Neurizon®'s lead investigational therapy for ALS * Clears the way for entry into the HEALEY ALS Platform Trial expected to commence in Q4 CY2025 * IND supported by robust preclinical safety data and comprehensive manufacturing and ...
Neurizon Enters into Global License with Elanco to Accelerate Commercialisation of NUZ-001
Highlights: * Neurizon® Therapeutics and Elanco Animal Health enter into an exclusive global licensing agreement * Provides worldwide rights for Neurizon to utilise Elanco's intellectual property, providing exclusive access to an extensive package of non-clinical studies and manufacturing da...
NUZ-001 Shows Promise in Zebrafish Model of Huntington's Disease
Highlights: * NUZ-001 and its active metabolite NUZ-001 Sulfone demonstrated significant neuroprotective effects in a zebrafish model of Huntington's disease * Treatment prevented hallmark developmental and morphological abnormalities, protected against neuronal cell death, restored delayed h...
Neurizon Files IND Application to Support HEALEY ALS Platform Trial
Highlights: * IND application submission to the U.S. Food and Drug Administration (FDA) is a pivotal step in initiating a Phase 2/3 clinical study for NUZ-001 * The IND is a comprehensive dossier of information, including animal and human studies, pharmacokinetic analyses, toxicology studies,...
Neurizon's NUZ-001 Reduces Aggregation of Key ALS Disease Target TDP-43 in Preclinical Study
Highlights: * Positive preclinical results in human in vitro iPSC Motor Neuron models of Amyotrophic Lateral Sclerosis (ALS) * Neurizon's lead drug NUZ-001 and its major active metabolite significantly and dose-dependently prevented the aggregation of TAR DNA-binding protein 43 (TDP-43) by ~...
Neurizon Therapeutics Receives Positive Opinion on Orphan Medicinal Product Designation for NUZ-001 in Europe
Highlights: * Notice of positive opinion received from the European Medicines Agency for Orphan Medicinal Product Designation for NUZ-001 in Amyotrophic Lateral Sclerosis * Orphan Designation offers 10 years of market exclusivity in Europe upon product approval, along with additional regulat...
PharmAust affirms corporate strategy with name change to Neurizon Therapeutics
MELBOURNE, Australia, Oct. 15, 2024 /PRNewswire/ -- Neurizon Therapeutics Limited (ASX: NUZ) ("Neurizon" or "the Company"), a clinical-stage biotech company dedicated to advancing treatments for neurodegenerative diseases, is pleased to announce it has officially changed its name from PharmAust L...