Brii Bio Publishes 2022 Environmental, Social and Governance Report
Brii Bio receives an "A" grade in the latest MSCI ESG Rating
DURHAM, N.C. and BEIJING, April 28, 2023 /PRNewswire/ -- Brii Biosciences
Limited
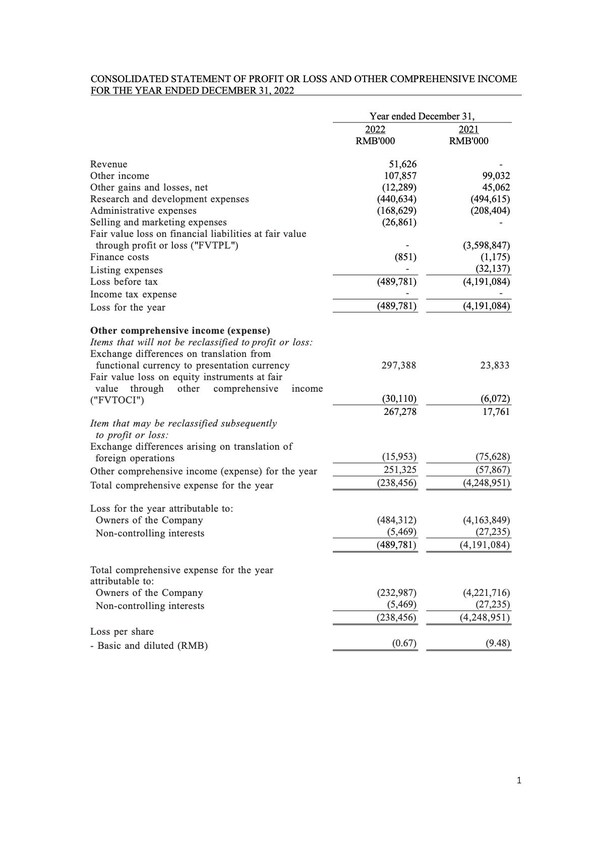
Brii Biosciences Provides Corporate Update and Reports Full-Year 2022 Financial Results
Company maintains priority focus on clinical programs to develop a novel f unctional cure for hepatitis B viral (HBV) infection and a potential first-of-its-kind treatment for postpartum depression (PPD) and major depressive disorders (MDD) Multiple Phase 2a proof-of-concept (POC) clinical data r...
Brii Biosciences Provides Update on Strategic Clinical Development Progress
Topline Phase 2 data from multiple hepatitis B viral (HBV) infection functional cure combination studies expected in the first half of 2023 Initiation of Phase 2 postpartum depression (PPD) study planned for early 2023 to advance potential first-of-its-kind treatment U.S. FDA lifted clinical hol...
Brii Biosciences Presents Promising Clinical Data to Support Ongoing Development of Novel HIV Therapeutic Candidates at IDWeek 2022
* Findings from two Phase 1 studies show both BRII-732 and BRII-778 are safe and well-tolerated * Results indicate potential for oral once-weekly, long-acting combination therapy for HIV infection, supporting continued clinical development * Additional data presented by Brii's licensing part...
Brii Biosciences Announces Top-line Results from Phase 1 Study of BRII-296, A Long-Acting Therapy in Development for Postpartum Depression
* Data demonstrated that a single administration of BRII-296 at 600 mg delivered a favorable pharmacokinetic profile and is safe and well-tolerated in healthy subjects * Findings suggest BRII-296 has potential to provide a safe and effective, one-time injectable treatment option to people wit...
Brii Biosciences Appoints Taiyin Yang to Board of Directors, Strengthening Product Quality Oversight to Support Company Momentum
Addition increases diversity of Board governance and supports further
independent expertise
DURHAM, N.C. and BEIJING, Sept. 1, 2022 /PRNewswire/ -- Brii Biosciences
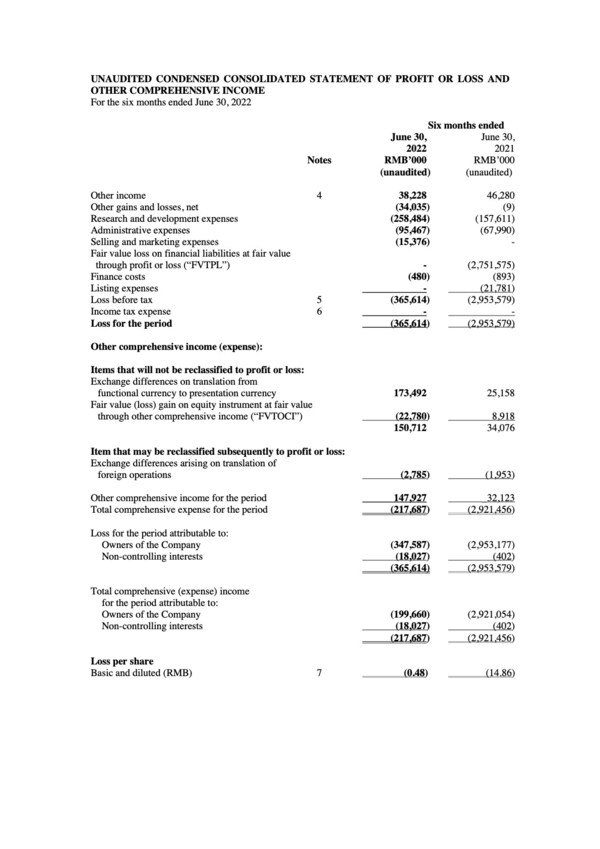
Brii Biosciences Provides Corporate Update and Reports 2022 Interim Results
Additions to executive team strengthen global leadership and position Company for strategic long-term growth First-ever product launch of long-acting amubarvimab/romlusevimab combination therapy for COVID-19 inChina advances Brii Bio from clinical development to commercial stage biotechnology co...
Brii Biosciences Appoints Eleanor de Groot as Chief Technology Officer and Aleksandar Skuban as Central Nervous System Diseases Therapy Area Head
Building a leadership team with global and local experience and expertise to
enable rapid execution across Company's broad therapeutic strategy
DURHAM, N.C. and BEIJING, Aug. 23, 2022 /PRNewswire/ -- Brii Biosciences
Brii Biosciences Announces Positive Data Demonstrating its Long-Acting COVID-19 Neutralizing Antibody Therapy, Amubarvimab/Romlusevimab Combination, Retains Neutralizing Activity Against Live Omicron Virus BA.4/5 and BA.2.12.1 Subvariants
Independent lab results demonstrate that the combination therapy retains neutralizing activity against currently dominant COVID-19 strains, as previously demonstrated with all variants of concern to date The amubarvimab/romlusevimab combination is commercially available in China and is under rev...
Brii Bio Announces Strategic Partnership with China Resources Pharmaceutical Commercial Group to Advance the Commercialization of Long-acting COVID-19 Neutralizing Antibody Therapy, Amubarvimab/Romlusevimab Combination, in China
The partnership will accelerate stockpiling, channel distribution and hospital access for the amubarvimab/romlusevimab combination in China The amubarvimab/romlusevimab combination is the first locally-discovered and approved SARS-CoV-2 target-specific treatment inChina, evaluated through a rand...
Brii Biosciences Appoints Dr. Susannah Cantrell as Chief Business Officer
Strategic executive hire adds to Company's global leadership team and positions
the Company for continued growth
DURHAM, N.C.and BEIJING, July 13, 2022 /PRNewswire/ -- Brii Biosciences
Limited
Brii Biosciences Announces Commercial Launch of its Amubarvimab/Romlusevimab Combination Therapy for COVID-19 in China
The Company is working closely with its commercial partners to supply the combination therapy to patients in need Brii Bio progressed the combination therapy from discovery to global Phase 3 data readout and first regulatory approval by the China NMPA in less than 20 months, validating the Com...
Brii Biosciences Exercises Option for Vir Biotechnology's VIR-3434, a Broadly Neutralizing Monoclonal Antibody Targeting Hepatitis B in Greater China
Exclusive development and commercialization rights to VIR-3434 in China strengthen Company's leadership and robust clinical pipeline in HBV Key partnership with Vir enables multitude of combination treatment options as part of Company's strategic approach to developing a functional cure for HBV ...
Brii Biosciences Announces Positive Data from a Randomized, Single-Blind Study of its Long-Acting COVID-19 Neutralizing Antibody Therapy, Amubarvimab/Romlusevimab Combination, in China
Data are consistent with the results of the global Phase 3 ACTIV-2 trial, demonstrating a favorable safety and tolerability profile in people with both severe and non-severe SARS-CoV-2 infections inChina Clinical benefits were observed in efficacy indicators such as RNA conversion rate, resoluti...