BrightGene Announces Positive Topline Phase 1 Results of Oral BGM0504 in Obesity in China and the United States
BGM0504 shown to be generally well tolerated Overall safety, pk, and efficacy profile supports once-daily oral dosing SUZHOU, China, March 25, 2026 /PRNewswire/ -- BrightGene Bio-Medical Technology Co., Ltd. (SSE: 688166, "BrightGene" or "the Company") today announced positive topline results f...
BrightGene Presents Positive Phase 2 Data for Dual GLP-1R/GIPR Agonist for Weight Management and Type 2 Diabetes and Preclinical Data for Novel Amylin Analog at American Diabetes Association's 85th Scientific Sessions
* Phase 2 data highlights best-in-class potential of dual GLP-1R/GIPR agonist BGM0504 for weight management and metabolic risk reduction in individuals with type 2 diabetes and overweight, non-obese individuals * Preclinical data for BGM1812 supports further development as next-generation amy...
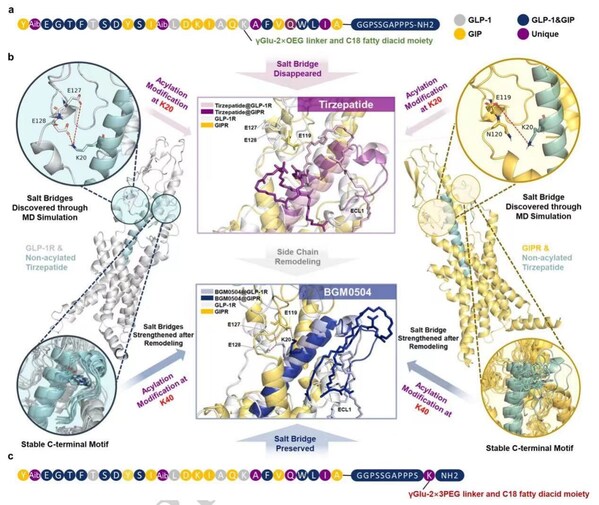
Nature Study Reveals: AI and Molecular Dynamics Designed BGM0504 Exhibits Superior Potency
SHANGHAI, July 22, 2024 /PRNewswire/ -- The molecular design strategy and experimental results of Bright Gene's dual GLP-1/GIP receptor agonist, BGM0504, have been published online in Scientific Reports, a sub-journal of Nature, on July 19, 2024. Bright Gene (Stock Code: 688166.SH) is an innovativ...