Biotechnology
ImmVira's Oncolytic Product MVR-T3011 Expanded to BCG-Naïve Bladder Cancer Patients for the First Time with Clinical Data Presented at the 2026 ASCO GU Conference
SUZHOU, China, Feb. 26, 2026 /PRNewswire/ -- Intravesical BCG is the standard of care (SOC) for BCG-naïve high-risk non-muscle-invasive bladder cancer (NMIBC) patients. However, the scarcity of BCG products has become a global phenomenon, and coupled with the side effects of BCG therapy itself,me...
Illumina Launches TruPath Genome, Unveils NovaSeq X Roadmap, and Drives Cancer Breakthroughs with Connected Multiomics
* TruPath™ Genome offers more complete genome, with the simplest sample to sequencer workflow, and the new AGBT data demonstrates its accuracy in detecting rare genetic diseases * 40% increase in output to 35 billion reads, up to Q70 quality scores, improved turnaround time, and staggered sta...
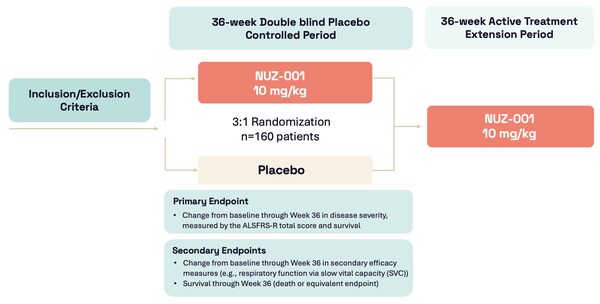
Neurizon Initiates Dosing of NUZ-001 in HEALEY ALS Platform Trial
Highlights: * First participant enrolled and dosed with NUZ-001 in Regimen I of the HEALEY ALS Platform Trial evaluating NUZ-001 for the treatment of ALS * Approximately 160 participants with ALS will be enrolled in a 36-week randomised,double-blind, placebo-controlled adaptive Phase 2/3 clin...
SK bioscience, IDT Biologika and Vaxxas Selected for Next-Generation Influenza Vaccine development Financed by the European Union
* SK–IDT–Vaxxas consortium establishes strategic entry point for high-density microarray patch (HD-MAP) vaccines inEurope to improve vaccine performance and access. * Parallel development of high-immunogenicity HD-MAP vaccines for seasonal and pandemic influenza threats. INCHEON, South Korea,...
CJ BIO Publishes Clinical Study on BiomeNrich™ POST SZ075, a Postbiotic Ingredient Demonstrating Skin Improvement Benefits
SEOUL, South Korea, Feb. 26, 2026 /PRNewswire/ -- CJ BIO announced the publication of clinical research findings on its postbiotic ingredient, BiomeNrich™ POST SZ075, in an international peer-reviewed journal. The study strengthens the scientific foundation of the ingredient and supports its pos...
genOway: Revolutionizing the Prediction of Antibody Therapies in Humans with Unique Mouse Model
LYON, France, Feb. 26, 2026 /PRNewswire/ -- Antibody therapies are changing the treatment of cancer, inflammatory diseases and infections. However, many promising drug candidates fail in clinical trials. A study in Science Immunology shows that genOway's genO-hFcγR mouse model enables more accura...
Illumina partners go beyond the genome, driving cancer breakthroughs using spatial transcriptomics, epigenomics, and proteomics
At AGBT, researchers reveal new findings from studies leveraging Illumina's multiomics solutions SAN DIEGO, Feb. 26, 2026 /PRNewswire/ -- Illumina, Inc. (NASDAQ: ILMN) announced new customer breakthroughs in oncology powered by Illumina's spatial transcriptomics, 5-base sequencing, and proteomi...
Discovery linking gut bacteria to cancer treatment wins the Bial Award in Biomedicine and earns €350,000 prize
A study published in Science reveals that a healthy gut microbiota can enhance the efficacy of immunotherapy used to treat cancer, while antibiotics may impair its effect by reducing intestinal microbiota diversity. PORTO, Portugal, Feb. 25, 2026 /PRNewswire/ -- An international consortium of 48...
Sanya Yazhou Bay Science and Technology City Appoints Intrinsic SEA as Southeast Asia Strategic Partner Following Landmark Hainan FTP Special Customs Operations
SANYA, China, Feb. 25, 2026 /PRNewswire/ -- Following the historic launch of island-wide independent customs operations in the Hainan Free Trade Port (FTP) onDecember 18, 2025, Sanya Yazhou Bay Science and Technology City (YZBSTC) has entered into a strategic partnership with Intrinsic SEA as its...
SteinCares and Shilpa Biologicals Strike into Licensing Agreement to Expand Access to Biosimilars Across Latin America
* New partnership combines Shilpa Biologicals' development and manufacturing capabilities with SteinCares' regional commercialization platform * Collaboration aims to expand patient access to cost-effective treatments and reinforces SteinCares' leadership in biosimilars across Latin America ...
Antengene Announces Clinical Collaboration with Junshi Biosciences to Explore the Synergistic Potential of ATG-037 (Oral CD73 Inhibitor) In Combination with JS207 (PD-1/VEGF BsAb)
SHANGHAI and HONG KONG, Feb. 25, 2026 /PRNewswire/ -- Antengene Corporation Limited ("Antengene", SEHK: 6996.HK) , a leading innovative, commercial-stage global biotech company dedicated to discovering, developing and commercializing first-in-class and/or best-in-class medicines for autoimmune d...
Direct Genome-Scale Mapping of Endonuclease Activity of the Human LINE-1 ORF2p Endonuclease
Nabsys and the Research Lab of Dr. Martin Taylor, Brown University, Present Data Using the OhmX™ Platform at AGBT 2026 PROVIDENCE, R.I., Feb. 25, 2026 /PRNewswire/ -- Nabsys 2.0, LLC (Nabsys), a pioneer in electronic genome mapping (EGM) technology, today announced new findings presented at the ...
Sanyou Biopharmaceuticals Announces New Strategic Investor; Liferiver Biotech Increases Its Stake, and Gree Group's Yanghe Investment Joins as a Strategic Shareholder
SHANGHAI, Feb. 24, 2026 /PRNewswire/ -- Sanyou Biopharmaceuticals (Shanghai) Co., Ltd. ("Sanyou Bio"), a biotech company powered by super-trillion-molecule libraries and AI-driven technologies, today announced the completion of a significant equity restructuring. The shares previously held by Hai...
Illumina unveils roadmap of groundbreaking NovaSeq X advancements in data quality, output, speed, and flexibility
40% increase in output to 35 billion reads, up to Q70 quality scores, improved turnaround time, and staggered starts are among the updates to be rolled out on all NovaSeq X systems, advancing precision medicine and delivering compounded value for NovaSeq X customers SAN DIEGO, Feb. 24, 2026 /PRN...
Sciwind Biosciences Partners with Pfizer China to Commercialize its Biased GLP-1 in China
HANGZHOU, China, Feb. 24, 2026 /PRNewswire/ -- Hangzhou Sciwind Biosciences Co., Ltd. ("Sciwind Biosciences") today announced a strategic commercialization collaboration with Pfizer China for Ecnoglutide injection (Ecnoglutide), a new–generation cAMP–biased GLP–1 receptor agonist. Under the agree...
"Would You Clone Yourself for a Cure?" Clonell Unveils White Paper on Fundamental Cures for 34 Incurable Diseases Using 'Human Cloning' Technology
Harnessing the vitality of a clone while neutralizing ethical controversies through a breakthrough 'Therapeutic Cloning' protocol—ushering in a new era of regenerative medicine to conquer cancer, Alzheimer's, and aging. YONGIN, South Korea, Feb. 23, 2026 /PRNewswire/ -- Human cloning has long be...
iRegene Hits Dual Global Firsts: U.S. Patient Dosed in Phase IIa for NouvNeu001 in Parkinson's and First Enrollment in China's Randomized MSA Trial for NouvNeu004
CHENGDU, China, Feb. 23, 2026 /PRNewswire/ -- The global race to treat neurodegenerative diseases has reached a new tempo as iRegene Therapeutics pushes its cell therapy pipeline into key clinical stages in the U.S. andChina. Concurrently, the company's "AI + Chemical Induction" platform saw its...
Formosa Pharmaceuticals Announces Licensing Agreement with Arrotex, for Commercialization of Clobetasol Propionate Ophthalmic Suspension for the Treatment of Inflammation and Pain Following Ocular Surgery
TAIPEI, Feb. 23, 2026 /PRNewswire/ -- Taiwan-based Formosa Pharmaceuticals ("Formosa", 6838.TW) announced today that the company has entered into an exclusive licensing agreement with Arrotex Pharmaceuticals Pty Ltd. ("Arrotex"), for exclusive rights to the commercialization of clobetasol propio...
CStone Announces MHRA Approval in UK for Sugemalimab in Stage III NSCLC
* Following approval by the European Commission (EC), sugemalimab has received a new indication approval from the UK Medicines and Healthcare products Regulatory Agency (MHRA) for stage III non-small cell lung cancer (NSCLC). This marks the second indication approved for the product in the UK. ...
Cambrex Announces Cornell Professor Song Lin as Recipient of the 2025 Snapdragon Prize for Innovation in Chemistry Technology
EAST RUTHERFORD, N.J., Feb. 19, 2026 /PRNewswire/ -- Cambrex, a leading global contract development and manufacturing organization (CDMO) and Snapdragon Chemistry, a Cambrex company, are proud to announce that Dr. Song Lin, Tisch University Professor in chemistry and chemical biology at Cornell U...
Week's Top Stories
Most Reposted
Visa and Trip.com Group Ink Strategic Collaboration, Enabling Consumers to Chase Their Passions Through Travel
[Picked up by 309 media titles]
2026-05-19 18:10Two-day Global Prosperity Summit 2026 officially opens
[Picked up by 308 media titles]
2026-05-19 23:59Two‑day Global Prosperity Summit 2026 concludes successfully
[Picked up by 303 media titles]
2026-05-21 22:13Hyundai Motor Group to Pioneer Hong Kong's Hydrogen Economy, Accelerating Asia-Pacific Expansion
[Picked up by 290 media titles]
2026-05-18 15:30Autonomous AI marketing platform Protaigé launches with Maia, the world's first AI Account Director that operates within the flow of work
[Picked up by 281 media titles]
2026-05-20 09:15