Biotechnology
Origin Agritech and China Agricultural University to Collaborate on Research of Molecular Design of Corn Using CRISPR
BEIJING, Sept. 21, 2021 /PRNewswire/ -- Origin Agritech Ltd. (NASDAQ: SEED) (the "Company" or "Origin"), an agriculture technology company, today announced that it has entered into collaboration agreement with China Agricultural University for the research of molecular design in corn. One of the...
Novavax to Participate in Fireside Chat at Devex @ UNGA 76
GAITHERSBURG, Md., Sept. 21, 2021 /PRNewswire/ -- Novavax, Inc. (Nasdaq: NVAX), a biotechnology company developing and commercializing next-generation vaccines for serious infectious diseases, today announced thatJohn J. Trizzino, Executive Vice President, Chief Commercial Officer and Chief Busin...
RhoVac announces the completion of patient recruitment for the BRaVac clinical phase IIb study of RV001 in prostate cancer
STOCKHOLM, Sept. 21, 2021 /PRNewswire/ -- RhoVac AB ("RhoVac"), a Swedish cancer immunotherapy company, announces today onSep 20th , 2021, that its clinical phase IIb study of lead drug candidate RV001 in prostate cancer, BRaVac, is deemed fully recruited and that no additional patients will be ...
Alterity Announces Presentation of Biomarker Data at the International Parkinson and Movement Disorder Society Congress 2021
MELBOURNE, Australia and SAN FRANCISCO, Sept. 20, 2021 /PRNewswire/ -- Alterity Therapeutics (ASX: ATH, NASDAQ: ATHE) ("Alterity" or "the Company"), a biotechnology company dedicated to developing disease modifying treatments for neurodegenerative conditions, today announced that data was present...
PharmAbcine to Participate in BIO-Europe 2021
DAEJEON, South Korea, Sept. 20, 2021 /PRNewswire/ -- PharmAbcine Inc. (KOSDAQ: 208340ks), a clinical-stage biotech company focusing on the development of next generation antibody therapeutics, announced today that the Company will virtually participate in the 27th annual BIO-Europe which will tak...
Bionomics Prepares BNC210 for Start of Phase 2 Acute Treatment of Social Anxiety Disorder Trial
* Rapid oral absorption of BNC210 novel tablet formulation potentially well-suited for acute treatment of anxiety in patients with Social Anxiety Disorder * Phase 2 clinical trial on target to start by end of 2021 and expected to read out topline data by end of 2022 ADELAIDE, Australia, Sept....
Terumo celebrates 100-year anniversary in Asia with an eye to the future
SINGAPORE, Sept. 20, 2021 /PRNewswire/ -- Terumo, a global leader in medical technology, celebrates its centenary inAsia. From humble beginnings as a thermometer manufacturer in 1921, and the first company in 1963 to create disposable syringes inJapan, the company has been providing medical solut...
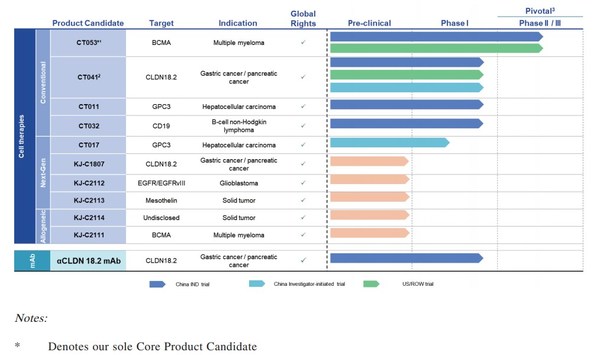
Appealing Data of CARsgen Therapeutics' CAR-T (CT041) in Advanced Gastric Cancer Presented at ESMO
SHANGHAI, Sept. 19, 2021 /PRNewswire/ -- On September 19, 2021, CARsgen Therapeutics (stock code: 2171.HK) disclosed the latest progress of the investigator-initiated trial (IIT) of Claudin18.2 (CLDN18.2) CAR-T (CT041) for the treatment of digestive system tumors. Results of this trial have been ...
Illumina reaffirms commitment to the National Project of Bio Big Data in Korea
MELBOURNE, Australia, Sept. 20, 2021 /PRNewswire/ -- Illumina, Inc. (NASDAQ: ILMN) today announced that the National Project of Bio Big Data in Korea has selected Illumina technology for the second pilot project in their effort to work towards establishing a national digital library on health and...
2021 CSCO | Henlius will Release Four Oral Presentations of Two Upcoming Commercial Products: Novel anti-PD-1 mAb Serplulimab and Bevacizumab Biosimilar
SHANGHAI, Sept. 18, 2021 /PRNewswire/ -- The 24th Annual Meeting of Chinese Society of Clinical Oncology (CSCO) will be held online and in-person from 25th to 29th September 2021. In this meeting, Henlius will release 4 study results of 2 products to be commercialized, the novel anti-PD-1 mAb ser...
Innovent Releases Interim Analysis Results of Sintilimab plus Chemotherapy for the First-Line Treatment of Esophageal Squamous Cell Carcinoma in the Phase 3 ORIENT-15 Study at ESMO Congress 2021
SAN FRANCISCO and SUZHOU, China, Sept. 18, 2021 /PRNewswire/ -- Innovent Biologics, Inc. ("Innovent") (HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures and commercializes high quality medicines for the treatment of cancer, metabolic, autoimmune and other major dis...
The NDA of Henlius Novel Anti-PD-1 mAb Serplulimab for First-Line Treatment of sqNSCLC Accepted by China's NMPA, Phase 3 MRCT Met its Primary Endpoint
SHANGHAI, Sept. 17, 2021 /PRNewswire/ -- Shanghai Henlius Biotech, Inc. (2696.HK) announced that the New Drug Application (NDA) of Serplulimab Injection (HLX10), a novel anti-PD-1 monoclonal antibody (mAb) independently developed by the company, in combination with carboplatin and albumin-bound ...
Global Cord Blood Corporation to Hold Annual General Meeting in Hong Kong on December 7, 2021
HONG KONG, Sept. 17, 2021 /PRNewswire/ -- Global Cord Blood Corporation (NYSE: CO) ("GCBC" or the "Company"),China's leading provider of cord blood collection, laboratory testing, hematopoietic stem cell processing, and stem cell storage services, today announced that the Company will hold its 20...
Zhongchao Inc. Launches Hematology MDT Training Platform to Improve Diagnosis and Treatment
SHANGHAI, Sept. 17, 2021 /PRNewswire/ -- Zhongchao Inc. (NASDAQ: ZCMD) ("Zhongchao" or the "Company"), an internet technology company offering healthcare professionals the online healthcare information, professional training and educational services platform and patients the patient management p...
Zhongchao Inc. Launches Hematology MDT Training Platform to Improve Diagnosis and Treatment
SHANGHAI, Sept. 17, 2021 /PRNewswire/ -- Zhongchao Inc. (NASDAQ: ZCMD) ("Zhongchao" or the "Company"), an internet technology company offering healthcare professionals the online healthcare information, professional training and educational services platform and patients the patient management p...
BioVaxys Cancer Vaccine Manufacturing Partner Bio Elpida Reaches Bioproduction Milestone
VANCOUVER, British Columbia, Sept. 17, 2021 /PRNewswire/ -- BioVaxys Technology Corp. (CSE: BIOV) (FRA: 5LB) (OTCQB: BVAXF) ("BioVaxys"), announced today that cancer vaccine manufacturing partner Bio Elpida inLyon, France, has reached major milestones in the development of the bioproduction proce...
Innovent Releases Phase 2 Results of Pemigatinib in Chinese Patients with Advanced Cholangiocarcinoma at ESMO Congress 2021
SAN FRANCISCO and SUZHOU, China, Sept. 17, 2021 /PRNewswire/ -- Innovent Biologics, Inc. ("Innovent", HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures and commercializes high-quality medicines for the treatment of cancer, metabolic, autoimmune and other major dise...
Arctic Vision Announces Addition of Novel Mydriatic Drug MydCombi™ (ARVN004) to Eyenovia Commercialization Deal
SHANGHAI, Sept. 16, 2021 /PRNewswire/ -- Arctic Vision, a China-based biotech company focused on innovative ophthalmic therapies, announced today that it will expand its exclusive license agreement with Eyenovia, Inc. (NASDAQ: EYEN) to include a third asset, MydCombi™, for development and commerc...
Bridge Biotherapeutics Presents Pre-Clinical Data of BBT-176 at the European Society for Medical Oncology (ESMO) Virtual Congress 2021
* Pre-clinical data exploring the in vitro and in vivo efficacy of BBT-176 will be disclosed via an ePoster at the ESMO Congress 2021 * Patient dosing for the phase 1/2 clinical study of BBT-176 was initiated in April 2021…the topline data of the first-in-human, dose escalation study is expec...
PharmaZell and Novasep enter into exclusive negotiations in new drive to create a technology-driven leader for complex small molecules and ADCs of global scale
RAUBLING, Germany and LYON, France, Sept. 17, 2021 /PRNewswire/ -- PharmaZell, the German producer of highly resilient and specialty APIs, andFrance-based Novasep, a leading CDMO focused on complex small molecules and ADCs have entered into exclusive negotiations to create a leading European plat...
Week's Top Stories
Most Reposted
Visa and Trip.com Group Ink Strategic Collaboration, Enabling Consumers to Chase Their Passions Through Travel
[Picked up by 309 media titles]
2026-05-19 18:10Two-day Global Prosperity Summit 2026 officially opens
[Picked up by 308 media titles]
2026-05-19 23:59Two‑day Global Prosperity Summit 2026 concludes successfully
[Picked up by 303 media titles]
2026-05-21 22:13Autonomous AI marketing platform Protaigé launches with Maia, the world's first AI Account Director that operates within the flow of work
[Picked up by 281 media titles]
2026-05-20 09:15Consistently recognised: Infobip named a Leader in Gartner® Magic Quadrant™ for CPaaS again
[Picked up by 279 media titles]
2026-05-21 16:31