Medical/Pharmaceuticals
Ivonescimab (PD-1/VEGF) Received Priority Review from China's NMPA for First-Line Treatment of PD-L1 Positive NSCLC
The Second Indication of Ivonescimab in NSCLC 2L+ EGFRm NSCLC Already Approved in May 2024 HONG KONG, Aug. 14, 2024 /PRNewswire/ -- Akeso, Inc. (HKEX: 9926.HK) ("Akeso,") today announced that the National Center for Drug Evaluation of the State Drug Administration ofthe People's Republic of China...
REMOVERY TO PROVIDE 150 FREE TATTOO REMOVAL PACKAGES TO COMMEMORATE THE INAUGURAL WORLD TATTOO REMOVAL DAY
On August 14, the world's tattoo removal experts will provide 150 treatments to individuals in need across their global studio locations AUSTIN, Texas, Aug. 14, 2024 /PRNewswire/ -- To bring awareness to the safety, efficacy and accessibility of tattoo removal treatment,Removery is presenting th...
Strategic Partnership Between Beyond Medical Group (Singapore) and SingMed Medical Centre (Cambodia) Enhances Healthcare Collaboration
Beyond Medical Pledges to Join Forces with SingMed to Enhance Access to
Healthcare and Elevate Care Standards Across Both Regions
SINGAPORE, Aug. 14, 2024 /PRNewswire/ -- Beyond Medical Group Singapore
Alfred Health Enhances Cancer Patient Care with Oracle Health
Leading Victorian healthcare network integrates oncology and EHR solutions to deliver more informed care across its locations AUSTIN, Texas and VICTORIA, Australia, Aug. 13, 2024 /PRNewswire/ -- Leading Victorian healthcare network, Alfred Health, has extended its long-standing Oracle Health foo...
Qilian International Holding Group Ltd. Announces Trading Ticker Symbol to "BGM"
CHENGDU, China, Aug. 13, 2024 /PRNewswire/ -- Qilian International Holding Group Ltd. ("Qilian" or the "Company") (NASDAQ: QLI), aChina-based pharmaceutical and chemical products manufacturer, announced today that effective onAugust 11, 2024, its Class A ordinary shares will begin trading on the...
Sinch AI Used to Safely Pre-Triage Patients
STOCKHOLM, Aug. 13, 2024 /PRNewswire/ -- Sinch (Sinch AB (publ) – XSTO: SINCH), which is pioneering the way the world communicates through its Customer Communications Cloud, is excited to announce that MINDD, a Dutch medical technology company, has selected Sinch to develop an advanced AI-driven...
Australian biotech incubator Proto Axiom announces $20M Series B first close
$20M unlocked for opportunities in Australian biotech innovation, valuing Proto Axiom at$90M SYDNEY, Aug. 13, 2024 /PRNewswire/ -- Proto Axiom, the Australian biomedical technology incubator has today announced a$20 million first close of its targeted$30 million Series B, to bolster Australia's...
Docquity Drives Demand for SIT Pharma's Duo Probiotics in Thailand, Achieving 31% Sales Growth in 6 months
* Docquity's Awareness to Advocacy (A2A) Program and strong local distribution network enabled SIT's duo probiotic sales to surpass the market growth rate of 15% * The technology platform boosted SIT's HCP education efforts, more than doubling its reach to 2,000 HCPs within six months and aim...
Registrational Phase III Study of Lisaftoclax in Newly-Diagnosed Patients with Higher-Risk Myelodysplastic Syndrome Cleared by China CDE
ROCKVILLE, Md. and SUZHOU, China, Aug. 13, 2024 /PRNewswire/ -- Ascentage Pharma (6855.HK), a global, integrated biopharmaceutical company engaged in discovering, developing and commercializing both first- and best-in-class therapies for malignancies, announced today it has been cleared by the Ce...
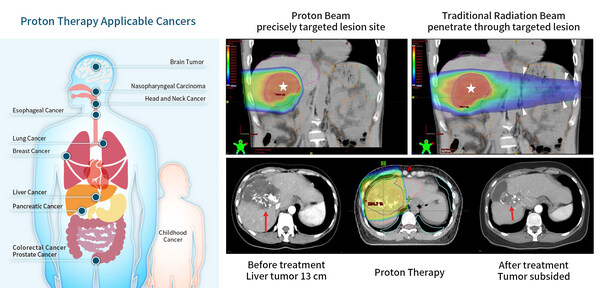
Chang Gung Cancer Proton Therapy Achieves Breakthrough
Research Confirms Effective Control of Large Liver Cancer, Doubling Survival
Time
Published in the International Journal of Radiation Oncology
Biosyngen's BRG01 Receives FDA Approval for Phase II Clinical Trial
SINGAPORE, Aug. 13, 2024 /PRNewswire/ -- Biosyngen is proud to announce that the U.S. Food and Drug Administration (FDA) has approved its BRG01, an EBV-specific CAR-T cell therapy, to proceed with a pivotal Phase lI clinical trial.This marks the first cell therapy to enter Phase lI trials in both...
Pantai Hospital Kuala Lumpur Introduces Outpatient Parenteral Antimicrobial Therapy (OPAT) for Improved Patient Care
KUALA LUMPUR, Malaysia, Aug. 12, 2024 /PRNewswire/ -- Pantai Hospital Kuala
Lumpur (PHKL) has officially introduced its Outpatient Parenteral Antimicrobial
Therapy (OPAT) service, offering an innovative treatment option that enhances
patient care.
AI in the IVF Process is the Standard of Care at Virtus Fertility Centre Singapore
The technology developed by Virtus Health's scientists contributes to IVF medicine by shortening the time to a successful pregnancy and improving overall success rates. SINGAPORE, Aug. 13, 2024 /PRNewswire/ -- Virtus Fertility Centre Singapore (VFCS) announced that artificial intelligence (AI), ...
MediPortal Introduces AI for Practice Management in Australia
PERTH, Australia, Aug. 13, 2024 /PRNewswire/ -- MediPortal PTY LTD. (MediPortal) has launched an AI-powered practice intelligence platform for Australian medical practices, aiming to enhance efficiency and patient care. Developed as the brainchild of a practising general practitioner, this innov...
Lynk Pharmaceuticals Announces Positive Results from Phase 1b Clinical Study of LNK01004 for the Treatment of Atopic Dermatitis
HANGZHOU, China and SHANGHAI and BOSTON, Aug. 13, 2024 /PRNewswire/ -- Lynk Pharmaceuticals Co., Ltd. (hereinafter referred to as "Lynk Pharmaceuticals"), a clinical-stage innovative drug development company, announced positive results from its Phase1b clinical study of LNK01004, a skin restricte...
Celltrion USA announces incorporation of adalimumab-aaty, a Humira® biosimilar, to the Costco Member Prescription Program
* Adalimumab-aaty was first approved by the Food and Drug Administration on May 23, 2023 and became commercially available among key distributors across the U.S. onJuly 2, 2023 * Adalimumab-aaty's inclusion creates greater accessibility to treatments for Americans with inflammatory conditions...
Environmental Compliance and Pharmaceutical Quality Standards Pave the Way for Transformational Growth in Chromatography
SAN ANTONIO, Aug. 13, 2024 /PRNewswire/ -- Chromatography instruments and modular test equipment are revolutionising scientific analysis and industrial testing. By enabling precise separation and analysis of complex mixtures, chromatography plays a crucial role in ensuring pharmaceutical purity, ...
OcuMension acquires rights to Alcon eye drops in exchange for a 16.7% equity stake, signaling the commencement of comprehensive strategic relationship
SHANGHAI, Aug. 12, 2024 /PRNewswire/ -- On August 13th 2024, Ocumension Therapeutics ("OcuMension") announced an agreement with Alcon Inc. ("Alcon", and collectively with OcuMension, the "Parties") where OcuMension will acquire or secure commercialization rights inChina to a portfolio of Alcon dr...
Mabwell's Novel Nectin-4 Targeting ADC 9MW2821 Granted Breakthrough Therapy Designation by China's NMPA
SHANGHAI, Aug. 12, 2024 /PRNewswire/ -- Mabwell (688062.SH), an innovative biopharmaceutical company with entire industry chain, announced its novel Nectin-4 targeting ADC (R&D code: 9MW2821) has been granted Breakthrough Therapy Designation (BTD) bythe Center for Drug Evaluation (CDE) of China's...
Ocumension (01477.HK) Demonstrates Strong Operational Resilience, Commercialization Capabilities Reach New Heights
SHANGHAI, Aug. 12, 2024 /PRNewswire/ -- On August 12, Ocumension Therapeutics ( Shanghai) Co., Ltd. (01477.HK, hereinafter referred to as "Ocumension" or the "Company") released its 2024 Interim Results Report. During the reporting period, the company achieved operating revenue ofRMB 168 million, ...
Week's Top Stories
Most Reposted
Marina Bay precinct partners UOB, Marina Bay Sands and Singapore Tourism Board, together with Disney Cruise Line, to illuminate Singapore's skyline with a fireworks sky show
[Picked up by 321 media titles]
2026-02-19 14:30NextFin Asia: A New Dedicated Fund for the Catapult: Inclusion SE Asia Program to Further Scale Inclusive Finance Fintechs in ASEAN
[Picked up by 301 media titles]
2026-02-23 08:00Little Artists Art Studio, Singapore Shines at Art Capital 2026
[Picked up by 280 media titles]
2026-02-17 19:12Vitafoods Asia 2026 Expands by 30%: A Bigger, More Dynamic Trade Event with Exciting New Features & Increased International Participation
[Picked up by 277 media titles]
2026-02-23 10:09Kung Fu Meets Spring -- Unitree Spring Festival Gala Robots Present "Cyber Real Kung Fu" in the Year of the Horse
[Picked up by 259 media titles]
2026-02-17 14:16