Medical/Pharmaceuticals
Pulnovo Medical Announces First Enrollment in Singapore for Global Clinical Study of PADN Technology in Treating Pulmonary Hypertension Associated with Left Heart Disease
SHANGHAI, Aug. 9, 2024 /PRNewswire/ -- Pulnovo Medical Limited,a globally recognized device pioneer in the treatment of Pulmonary Hypertension (PH) and Heart Failure (HF), is pleased to announce the launch ofAsia's first international multi-center clinical study of Percutaneous Pulmonary Artery ...
T-MAXIMUM PHARMACEUTICAL Announces Latest Clinical Advances in Allogeneic CAR-T Cell Therapy Breakthrough for Solid Tumors
Summary * T-Maximum Pharmaceutical develops allogeneic CAR-T therapies with a unique CRISPR/Cas9 Gene-Editing platform for solid tumors, provide a solution of two major pain points, GvHD and HvG, aiming to revolutionize cancer. * MT027, the product of target B7H3 received US FDA orphan drug d...
MALAYSIA HEALTHCARE TRAVEL COUNCIL (MHTC) CONTINUES TO ENHANCE AND ELEVATE THEIR "EXPERIENCE MALAYSIA HEALTHCARE (EMH) STORIES" SERIES WITH EPISODE 3
KUALA LUMPUR, Malaysia, Aug. 8, 2024 /PRNewswire/ -- Malaysia Healthcare Travel Council (MHTC) continues their ongoing efforts to enhance and elevate their "Experience Malaysia Healthcare (EMH) Stories" with the third episode featuring Prof. Dato Sri Dr.Alwi Mohamed Yunus from Institut Jantung Ne...
WuXi Biologics and Medigene Enter into a Research Collaboration for Off-the-Shelf TCR-Guided T Cell Engagers
* Partnership leverages Medigene's leadership for T cell receptor (TCR) generation and characterization and WuXi Biologics' unique anti-CD3 mAb, its T cell engager (TCE) platform and proprietary bispecific antibody platform WuXiBody™ * Collaboration is a three-year and potentially multi-TCR p...
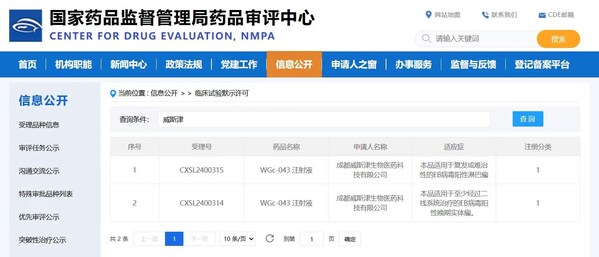
WestGene to Advance Clinical Trials Following Dual IND Approvals for World's First EB Virus-Related mRNA Therapeutic Cancer Vaccine
CHENGDU, China, Aug. 7, 2024 /PRNewswire/ -- WestGene Biopharma is proud to announce that its mRNA therapeutic cancer vaccine, WGc-043, has received dual IND approvals fromChina's National Medical Products Administration (NMPA) and the US FDA. This unprecedented achievement marks the world's firs...
Fierce Competition and Technological Innovation Drive Diabetes Care Devices Market Towards Explosive Growth
OEMs Leading the Charge with AI-Equipped Technology and Novel Business Strategies SAN ANTONIO, Aug. 8, 2024 /PRNewswire/ -- The diabetes care devices industry is witnessing a dynamic evolution, marked by intense competition among established players and emerging start-ups. Original Equipment Man...
PeproMene Bio announced study demonstrating strong polyfunctionality of novel chimeric antigen receptor (CAR) T cells
Polyfunctionality correlates with a patient's remission lasting over 18 months with the CAR T cell therapy IRVINE, Calif., Aug. 7, 2024 /PRNewswire/ -- PeproMene Bio, Inc., a clinical-stage biotech company developing novel therapies for cancers and immune disorders, today announced that City of...
Concord Medical Regains Compliance with NYSE Minimum Price Requirement
BEIJING, Aug. 7, 2024 /PRNewswire/ -- Concord Medical Services Holdings Limited ("Concord Medical" or the "Company") (NYSE: CCM), a healthcare provider specialized in cancer treatment, research, education and prevention inChina, today announced that it has received a letter from the New York Stoc...
Formosa Pharma and Eyenovia Announce Initiation of Co-Development of Clobetasol Propionate Ophthalmic Suspension, 0.05%, for the treatment of Acute Dry Eye Disease in United States
TAIPEI, Aug. 7, 2024 /PRNewswire/ -- Taiwan-based Formosa Pharmaceuticals ("Formosa", 6838.TWO) announced today that the company has signed a non-binding terms agreement with Eyenovia, Inc. ("Eyenovia", NASDAQ: EYEN), whereby the companies will co-develop Clobetasol Propionate Ophthalmic Suspensi...
I-Mab Appoints U.S. Auditor, PricewaterhouseCoopers LLP (PwC)
Engagement is part of I-Mab's commitment to transition to a U.S.-based biotech PwC to serve as independent registered public accounting firm for FY 2024 ROCKVILLE, Md., Aug. 7, 2024 /PRNewswire/ -- I-Mab (NASDAQ: IMAB) ("I-Mab", the "Company"), a U.S.-based, global biotech company, exclusively f...
Docquity Drives Demand for SIT Pharma's Duo Probiotics in Thailand, Achieving 31% Sales Growth in 6 months
* Docquity's Awareness to Advocacy (A2A) Program and strong local distribution network enabled SIT's duo probiotic sales to surpass the market growth rate of 15% * The technology platform boosted SIT's HCP education efforts, more than doubling its reach to 2,000 HCPs within six months and aim...
GSK's Jemperli (dostarlimab) plus chemotherapy approved in Singapore as the first frontline immuno-oncology treatment for dMMR/MSI-H primary advanced or recurrent endometrial cancer
* Jemperli in combination with chemotherapy is the first immuno-oncology treatment approved in the frontline setting for this patient population in Singapore.[1,2] * Latest published local registry reported endometrial cancer as the fourth most common cancer and had the ninth highest cancer mo...
Senhwa Biosciences receives US FDA Study May Proceed letter for the Phase I/II study of Silmitasertib (CX-4945) in combination with chemotherapy in children and young adults with relapsed refractory solid tumors
TAIPEI and SAN DIEGO, Aug. 6, 2024 /PRNewswire/ -- Senhwa Biosciences, Inc. (TPEx: 6492), a drug development company focusing on first-in-class therapeutics for oncology, rare diseases, and infectious diseases, today announced receipt of a "Study May Proceed" letter from the U.S. Food and Drug A...
Datasea's Digital Technology Subsidiary Selected as Prospective Partner for Future Projects with Subsidiary of China Mobile
The Prospective Partnership with a Subsidiary of China Mobile, one of the World's Largest Mobile Operator, Underscores DTSS' Achievements in 5G-AI Communications BEIJING, Aug. 6, 2024 /PRNewswire/ -- Datasea Inc. (Nasdaq: DTSS) ("Datasea" or the "Company"), a digital technology company incorpora...
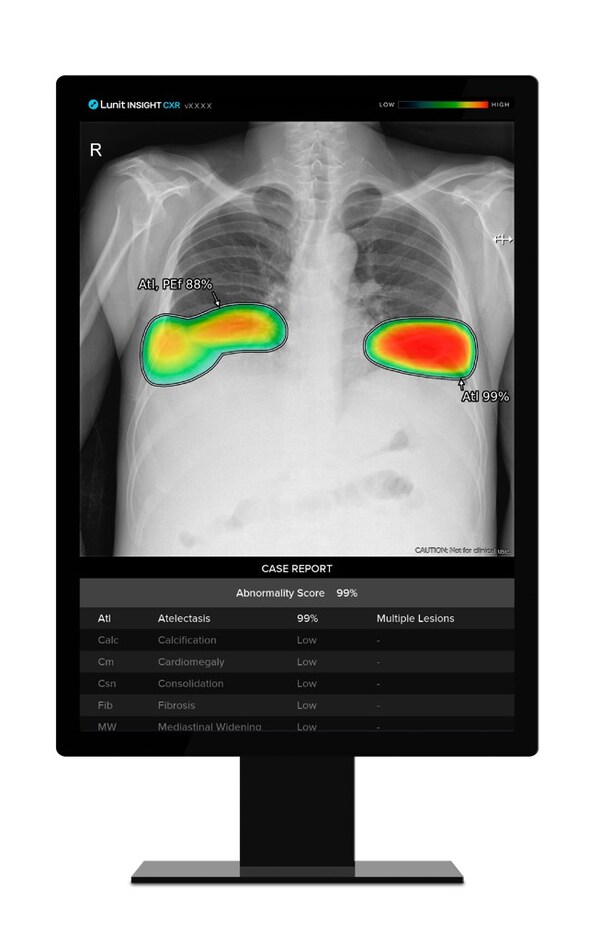
Lunit's AI for Tuberculosis Detection Tops in 12-Product Comparative Study
* Lancet Digital Health study highlights Lunit INSIGHT CXR as top performer in TB screening, showing promise for improving detection in high-burden settings SEOUL, South Korea, Aug. 6, 2024 /PRNewswire/ -- Lunit, a leading provider of AI-powered solutions for cancer diagnostics and therapeutics,...
PHYLEX VACCINE ON TRACK TO COMBAT DEADLY NIPAH VIRUS WITH POSITIVE RESULTS OF IMMUNOGENICITY STUDY
DEL MAR, Calif., Aug. 6, 2024 /PRNewswire/ -- Phylex Biosciences has achieved positive results with its new mRNA nanoparticle vaccine against the Nipah virus in an immunogenicity study conducted in collaboration with scientists of the U.S. Centers for Disease Control and Prevention (CDC) inAtlant...
Samsung Biologics joins the Pharmaceutical Supply Chain Initiative as Supplier Partner
* Samsung Biologics to embed PSCI principles into business practices for responsible value chain management * Supplier Partnership reaffirms company's commitment to decarbonize and build resilient supply chains INCHEON, South Korea, Aug. 6, 2024 /PRNewswire/ -- Samsung Biologics (KRX: 207940....
Latest Updates of Viva Biotech's Portfolio Companies
HONG KONG, Aug. 6, 2024 /PRNewswire/ -- Even with the ever-changing situation, technological innovation is still the most critical component for biopharmaceutical companies' long-term development. This continuous innovation keeps companies up to date and promotes the evolution of R&D and the succ...
First patient enrollment in the U.S. - A milestone for the global multi-center phase Ⅲ clinical trial of telitacicept for myasthenia gravis
YANTAI, China, Aug. 6, 2024 /PRNewswire/ -- RemeGen Co. Ltd.
The Adecco Group: Q2 2024 Results
Further market share gains and disciplined cost management; G&A savings above-target ZURICH, Aug. 6, 2024 /PRNewswire/ -- * Revenues -2% yoy in challenging markets; Group relative revenue growth +375 bps and Adecco GBU +220 bps * Revenues by GBU, Adecco -2% yoy, Akkodis -2% yoy, LHH -7% yoy ...
Week's Top Stories
Most Reposted
Marina Bay precinct partners UOB, Marina Bay Sands and Singapore Tourism Board, together with Disney Cruise Line, to illuminate Singapore's skyline with a fireworks sky show
[Picked up by 328 media titles]
2026-02-19 14:30Never Miss a Message: Agoda's Customer Support Now Travels With You
[Picked up by 326 media titles]
2026-02-24 12:00NextFin Asia: A New Dedicated Fund for the Catapult: Inclusion SE Asia Program to Further Scale Inclusive Finance Fintechs in ASEAN
[Picked up by 311 media titles]
2026-02-23 08:00Klook and Osaka Convention & Tourism Bureau sign MoU to advance inbound tourism and foster socio-economic development throughout Osaka Prefecture
[Picked up by 301 media titles]
2026-02-24 16:13Vitafoods Asia 2026 Expands by 30%: A Bigger, More Dynamic Trade Event with Exciting New Features & Increased International Participation
[Picked up by 288 media titles]
2026-02-23 10:09