Pharmaceuticals
Senhwa Biosciences Announces Abstract Accepted for Presentation at the 2022 ASCO Gastrointestinal Cancers Symposium
TAIPEI and SAN DIEGO, Nov. 15, 2021 /PRNewswire/ -- Senhwa Biosciences, Inc. (TPEx: 6492), a drug development company focusing on first-in-class therapeutics for oncology, rare diseases, and novel coronaviruses, announced that an abstract highlighting clinical design for their lead drug candidate...
Qilian International Holding Group Limited Announces Initiation of Development of New Drug for Gastric Cancer Prevention
JIUQUAN, China, Nov. 15, 2021 /PRNewswire/ -- Qilian International Holding
Group Limited (Nasdaq: QLI
RedHill Biopharma Announces Last Patient Randomized in Part A of Ongoing Phase 2/3 COVID-19 Study of Once-Daily Oral RHB-107 in Non-Hospitalized Patients
- Recruitment completed for Part A of the Phase 2/3 study of once-daily orally-administered RHB-107 (upamostat) for patients with symptomatic COVID-19 who do not require hospital care - Top-line results for Part A of the study, designed to evaluate safety and tolerability of RHB-107 and dose sel...
Seegene Opens New Subsidiary in Bogotá, Colombia, to Advance Regional COVID-19 Testing
SEOUL, South Korea, Nov. 15, 2021 /PRNewswire/ -- Seegene Inc. (KQ 096530), a leading biotechnology firm specializing in molecular diagnostics (MDx), has announced the opening of an eighth global subsidiary, strategically located in Bogotá,Colombia. The new entity, known as 'Seegene Colombia S.A....
Gannex Announces China NMPA Approved Phase II and III Protocols of ASC42, an FXR Agonist, for Treatment of Primary Biliary Cholangitis
SHANGHAI, Nov. 15, 2021 /PRNewswire/ -- Gannex Pharma Co., Ltd., a wholly owned company of Ascletis Pharma Inc. (HKEX:1672) announces today that the protocols of Phase II and III clinical trials of ASC42 to treat patients with primary biliary cholangitis (PBC) has been approved by China National ...
Innovent Announces the Results of the Phase Ib Clinical Trial of IBI302, a First-in-class Anti-VEGF and Anti-Complement Bi-specific Fusion Protein for Neovascular Age-Related Macular Degeneration at 2021 American Academy of Ophthalmology
SAN FRANCISCO and SUZHOU, China, Nov. 15, 2021 /PRNewswire/ -- Innovent Biologics, Inc. ("Innovent") (HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures and commercializes high-quality medicines for the treatment of oncology, metabolic, autoimmune and other major di...
Treadwell Therapeutics Announces A Presentation at the 2021 SITC Annual Meeting Featuring a Clinical Trial Update on CFI-402411, a First-in-Class HPK1 inhibitor
NEW YORK, Nov. 12, 2021 /PRNewswire/ -- Treadwell Therapeutics, a clinical-stage biotechnology company developing novel medicines for highly aggressive cancers, today announced a presentation for the Company's CFI-402411 program, an oral, first-in-class inhibitor ofHematopoietic Progenitor Kinase...
Kintor Pharma Announces Dosing of First Patient in Phase II Clinical Trial of KX-826 for the Treatment of Androgenic Alopecia Female Patients in China
SUZHOU, China, Nov. 12, 2021 /PRNewswire/ -- Kintor Pharmaceutical Limited ("Kintor Pharma", HKEX: 9939), a clinical-stage biotechnology company developing innovative small molecule and biological therapeutics, today announced that the company has dosed the first patient in its phase II clinical ...
InnoCare to Attend Upcoming Investor Conferences to Share Latest Company Development
BEIJING, Nov. 12, 2021 /PRNewswire/ -- InnoCare Pharma (HKEX: 09969), a leading biotech company, announced today that the company executives will attend the upcoming investor conferences organized by Goldman Sachs and Morgan Stanley to share company's latest development. Details are as follows: ...
The New Era Biotechnology deepens cooperation between China and Belarus in the Great Stone China Belarus Industrial Park (CBIP)
MINSK, Belarus, Nov. 12, 2021 /PRNewswire/ -- As an enterprise in CBIP, the New
Era Biotechnology Co., Ltd. participated in the fourth edition of the China
International Import Expo (CIIE) which opened inShanghai on November 5, 2021.
Hetero and Shenzhen Beimei Pharmaceuticals launch antiviral drug Oselavir® in China
Oseltamivir phosphate is used in the treatment of acute uncomplicated influenza infection in adults, adolescents and children BEIJING, Nov. 12, 2021 /PRNewswire/ -- Hetero, a globally renowned vertically integrated pharmaceutical organization and Shenzhen Beimei Pharmaceuticals jointly announced...
Recbio Announces Promising Preliminary Results From First-in-Human Study of a COVID-19 Vaccine Candidate With Novel Adjuvant BFA03
* Well tolerated and good safety profile, no SAE or TEAE leading to early discontinuation, no abnormal vital signs/laboratory testing results with clinical significance * 20μg ReCOV induced high titer of anti-SARS-CoV-2 neutralizing antibodies, with at least comparable level than published da...
PNOC Study in Childhood Brain Cancer Enrols First Patient
SYDNEY, Nov. 11, 2021 /PRNewswire/ -- Kazia Therapeutics Limited (NASDAQ: KZIA; ASX: KZA), an oncology-focused drug development company, is pleased to announce that PNOC022 (NCT05009992), a multi-drug phase II study in DIPG and diffuse midline gliomas, has been initiated at theUniversity of Calif...
Gmax's GMA106, second generation obesity/T2DM/NASH mAb gives first in human dose
HANGZHOU, China, Nov. 11, 2021 /PRNewswire/ -- Gmax Biopharm today announces that the first dose of GMA106 was given to human subjects in a phase 1 study to investigate the safety, pharmacokinetics, and pharmacodynamics of this drug in the treatment of obesity. The study is a single dose, placeab...
Everest Medicines and Gilead Sciences Jointly Announce Phase 2b Study of Sacituzumab Govitecan Conducted in China of Patients With Metastatic Triple-negative Breast Cancer Meets Primary Overall Response Rate Endpoint
SHANGHAI, Nov. 11, 2021 /PRNewswire/ -- Everest Medicines and Gilead Sciences, Inc. (Nasdaq: GILD) jointly announces today that Everest Medicines sponsored (HKEX 1952.HK) Phase2b EVER-132-001 study of sacituzumab govitecan (marketed as Trodelvy® in the United States) met its primary endpoint o...
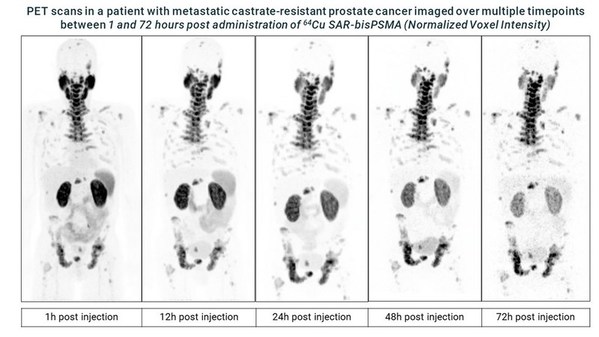
Recruitment for the dosimetry phase of Clarity's Cu-64/Cu-67 SAR-bisPSMA theranostic prostate cancer trial completed
* Clarity Pharmaceuticals completes recruitment for the initial dosimetry phase of its SAR-bisPSMA theranostic clinical trial SECuRE (NCT04868604)[1] investigating Targeted Copper Theranostics (TCT) in patients with metastatic castrate-resistant prostate cancer (mCRPC). * Dosimetry data is be...
IASO Biotherapeutics and Innovent Biologics to Present New BCMA CAR-T Cell Therapy Data in Oral Presentation at ASH 2021
SAN JOSE, Calif., NANJING, China and SHANGHAI, Nov. 10, 2021 /PRNewswire/ --
IASO Biotherapeutics("IASO Bio")
I-Mab and Jumpcan Announce Strategic Commercial Partnership on Eftansomatropin Alfa
* The partnership brings together the strengths of an innovative global biotech and a domestic leading pharmaceutical company specialized in and committed to pediatric medicines to accelerate the commercialization of eftansomatropin alfa * Jumpcan will pay I-Mab for a total of up to RMB 2.016...
Hummingbird Bioscience Announces Trials in Progress Poster Presentation at the Society for Immunotherapy of Cancer 2021 36th Annual Meeting
HOUSTON, Nov. 10, 2021 /PRNewswire/ -- Hummingbird Bioscience, an innovative clinical-stage biotech company focused on developing precision therapies against hard-to-drug targets, today announced a Trials in Progress poster presentation outlining the Phase 1 clinical trial design for HMBD-002, a ...
Ascletis Announces Approval of Phase II Clinical Trial of ASC22 (Envafolimab) by China NMPA for Immune Restoration/Functional Cure of HIV-1 Infected Patients
HANGZHOU, China and SHAOXING, China, Nov. 10, 2021 /PRNewswire/ -- Ascletis Pharma Inc. (HKEX: 1672) today announces the approval of the Phase II clinical trial of ASC22 (Envafolimab) by China National Medical Products Administration (NMPA) for the proposed new indication of immune restoration/fu...
Week's Top Stories
Most Reposted
Visa and Trip.com Group Ink Strategic Collaboration, Enabling Consumers to Chase Their Passions Through Travel
[Picked up by 309 media titles]
2026-05-19 18:10Two-day Global Prosperity Summit 2026 officially opens
[Picked up by 308 media titles]
2026-05-19 23:59Two‑day Global Prosperity Summit 2026 concludes successfully
[Picked up by 303 media titles]
2026-05-21 22:13Autonomous AI marketing platform Protaigé launches with Maia, the world's first AI Account Director that operates within the flow of work
[Picked up by 281 media titles]
2026-05-20 09:15AIMX Singapore 2026 Anchors Strategic Relationship with TechInnovation and AI Gravity
[Picked up by 279 media titles]
2026-05-19 14:00