Pharmaceuticals
Sana Biotechnology, IASO Biotherapeutics, and Innovent Biologics Announce Non-Exclusive License Agreement for Clinically Validated BCMA CAR Construct
SEATTLE and SAN FRANCISCO and SAN JOSE, Calif. and NANJING, China and SUZHOU, China andSHANGHAI, Jan. 10, 2022 /PRNewswire/ -- Sana Biotechnology, Inc. (NASDAQ: SANA), a company focused on creating and delivering engineered cells as medicines, IASO Biotherapeutics ("IASO Bio"), a clinical-stage ...
CARsgen Announces CT041 CAR T-cell Product Candidate Granted RMAT Designation by the FDA
SHANGHAI, Jan. 10, 2022 /PRNewswire/ -- CARsgen Therapeutics Holdings Limited (Stock Code: 2171.HK), a company mainly focused on innovative CAR T-cell therapies for the treatment of hematologic malignancies and solid tumors, today announced thatthe United States Food and Drug Administration (FDA)...
KAZIA THERAPEUTICS TO PRESENT AT HC WAINWRIGHT BIOCONNECT CONFERENCE
SYDNEY, Jan. 7, 2022 /PRNewswire/ -- Kazia Therapeutics Limited (NASDAQ: KZIA; ASX: KZA), an oncology-focused drug development company, is pleased to announce that CEO, DrJames Garner, will be speaking at the HC Wainwright BioConnect Conference, to be held virtually from10-13 January 2022. The co...
InnoCare to Attend Upcoming Investor Conferences to Share Latest Company Development
BEIJING, Jan. 7, 2022 /PRNewswire/ -- InnoCare Pharma (HKEX: 09969), a leading biotech company, announced today that the company executives will attend the upcoming investor conferences organized by Morgan Stanley and UBS to share company's latest development. Details are as follows: Morgan Stan...
LISCure Biosciences announces research collaboration with Mayo Clinic for rare liver diseases
SEOUL, South Korea, Jan. 6, 2022 /PRNewswire/ -- LISCure Biosciences Inc., a leading biotech focused on microbial-based therapeutics, announced that it has executed a research collaboration agreement with Mayo Clinic for new drug development for rare liver diseases. LISCure has been conducting j...
I-Mab Announces Senior Management Team Share Purchase Plan
SHANGHAI and GAITHERSBURG, MD., Jan. 6, 2022 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB), a clinical-stage biopharmaceutical company committed to the discovery, development, and commercialization of novel biologics, today announced that the senior management team of the Company, includin...
AKESO'S CADONILIMAB (PD-1/CTLA-4 BI-SPECIFIC ANTIBODY) COMBINED WITH IVONESCIMAB (PD-1/VEGF BI-SPECIFIC ANTIBODY) COMBINED WITH OR WITHOUT CHEMOTHERAPY OBTAINED APPROVAL TO INITIATE A PHASE Ib/II CLINICAL TRIAL FOR THE TREATMENT OF ADVANCED NON-SMALL CELL LUNG CANCER
HONG KONG, Jan. 6, 2022 /PRNewswire/ -- Today, Akeso, Inc. (09926.HK) announces that Cadonilimab (PD-1/CTLA-4 bi-specific antibody), the first-inclass novel immuno-oncology drug independently developed by the Company, combined with Ivonescimab (PD-1/VEGF bi-specific antibody), the novel immuno-o...
Nuance Pharma acquires Sino Health to further strengthen its commercial capabilities and expand pipelines
SHANGHAI, Jan. 6, 2022 /PRNewswire/ -- Nuance Pharma ("Nuance"or the "Company"), a specialty care focused biopharma with late late-stage clinical programs and existing commercial operations, today announced an agreement with Sino Health Pharmaceutical Limited ("Sino Health") , an affiliated compa...
Ascentage Pharma to Present at the J.P. Morgan 40th Annual Healthcare Conference
SUZHOU, China and ROCKVILLE, Md., Jan. 5, 2022 /PRNewswire/ -- Ascentage Pharma (6855.HK), a global biopharmaceutical company engaged in developing novel therapies for cancers, chronic hepatitis B (CHB), and age-related diseases, today announced its participation in and presentation at the 40th A...
Adlai Nortye Announces First Patient Dosed in Phase I Clinical Trial of the orally available, small-molecule PD-L1 inhibitor AN4005 for Advanced Tumors
NORTH BRUNSWICK, N.J. and HANGZHOU, China, Jan. 5, 2022 /PRNewswire/ -- Adlai Nortye Ltd. ("Adlai Nortye"), a clinical-stage biopharmaceutical company focused on the development of innovative cancer therapies, announced that the first patient was dosed in the Phase I clinical trial in the U.S. to...
Alterity Therapeutics to Present at the H.C. Wainwright BIOCONNECT Virtual Conference
MELBOURNE, Australia, Jan. 5, 2022 /PRNewswire/ -- Alterity Therapeutics (ASX: ATH, NASDAQ: ATHE) ("Alterity" or "the Company"), a biotechnology company dedicated to developing disease modifying treatments for neurodegenerative diseases, today announced thatDavid Stamler, M.D., Chief Executive Of...
PRISM BioLab, enters multi-project drug discovery collaboration with Roche and Genentech
TOKYO, Jan. 4, 2022 /PRNewswire/ -- PRISM BioLab, a Japan based biotechnology company with a proprietary peptide mimetic small molecule drug discovery platform, today announced it has entered into a multi-target research collaboration and licensing agreement with Roche and Genentech, a member of ...
Harbour BioMed Announces Dosing of First Patient of Two Phase I Trials for Next-Generation Anti-CTLA-4 Antibody HBM4003
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, Jan. 4, 2022 /PRNewswire/ -- Harbour BioMed ("HBM", HKEX: 02142) announced that, its next generation anti-CTLA-4 fully human heavy-chain only antibody (HCAb), HBM4003, has completed the first dosing of first patient in two phase I tri...
AKESO'S CADONILIMAB (PD-1/CTLA-4 BI-SPECIFIC ANTIBODY) COMBINED WITH CONCURRENT CHEMORADIOTHERAPY OBTAINED APPROVAL TO INITIATE A PHASE III CLINICAL TRIAL FOR THE TREATMENT OF LOCALLY ADVANCED CERVICAL CANCER
HONG KONG, Jan. 3, 2022 /PRNewswire/ -- Today, Akeso (09926.HK) announces that Cadonilimab (PD-1/ CTLA-4 bi-specific antibody), the first-inclass novel immuno-oncology drug independently developed by the Company, combined with concurrent chemoradiotherapy obtained approval from the Center for Dru...
Asieris Announces the World's First Patient Dose Administered in Combination of Asieris'APL-1202 and BeiGene's Tislelizumab as Neoadjuvant Therapy for MIBC Patients
SHANGHAI, Jan. 3, 2022 /PRNewswire/ -- Asieris Pharmaceuticals (Asieris), a global innovative pharma company specializing in new drugs for the treatment of genitourinary tumors, today announced that the world's first patient dose has been administered in U.S. for its oral APL-1202 in combination ...
Biosion, Inc. Appoints Anthony Yeh, Ph.D., as Chief Strategy Officer and Head of China BD
NEWARK, Del. and NANJING, China, Jan. 3, 2022 /PRNewswire/ -- Biosion, Inc. ("Biosion"), a global clinical stage biotechnology company, today announced the appointment of Dr.Anthony Yeh to the position of Chief Strategy Officer and Head ofChina business development (BD), where he will lead corpor...
I-Mab Announces Upcoming Participation at January Conferences
SHANGHAI and GAITHERSBURG, MD., Dec. 31, 2021 /PRNewswire/ -- I-Mab (the "Company") (Nasdaq: IMAB), a clinical-stage biopharmaceutical company committed to the discovery, development, and commercialization of novel biologics, today announced its participation in the following conferences inJanuar...
China Medical System (867.HK)'s Latest MSCI ESG Rating Unchanged at AA, Leading the Industry Globally
HONG KONG, Dec. 30, 2021 /PRNewswire/ -- Recently, Morgan Stanley Capital International (MSCI), the world's largest index provider, has updated the Environmental, Social and Governance (ESG) rating of China Medical System Holdings Limited ("CMS" or the "Company").CMS's latest rating remains uncha...
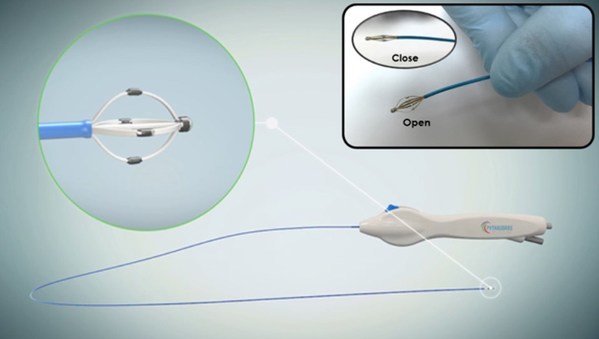
SyMap Medical Ltd. Announces its Strategic Collaboration with Pythagoras Medical
SUZHOU, China, Dec. 29, 2021 /PRNewswire/ -- SyMap Medical Ltd (SyMap) announced its strategic collaboration with Pythagoras Medical. With this win-win collaboration, SyMap has full rights to utilize all assets of Pythagoras, including their Renal Nerve Mapping System (ConfidenHT™, CE Mark Appro...
EOFlow and Zihipp Announce Joint Venture for Development of Innovative Solutions for Treatment of Obesity & NASH
* Establishment of Joint Venture Company named SanPlena in the US * Drug-device combination of EOFlow's smart wearable drug delivery platform and novel peptide derivatives developed by a team led by Prof. SirStephen Bloom , world-renowned researcher in the field of obesity and metabolism at Imp...
Week's Top Stories
Most Reposted
Digital Identity Co. Modernizes Thailand Immigration Bureau Services with AWS
[Picked up by 313 media titles]
2026-05-28 15:06The Philippines Sets a New Standard in Muslim-Friendly Tourism
[Picked up by 290 media titles]
2026-05-26 10:30ASEAN Business Advisory Council (ASEAN-BAC) and AVPN Establish Regional Platform to Strengthen Corporate Social Investment and Cross-Sector Collaboration Across Southeast Asia
[Picked up by 288 media titles]
2026-05-28 10:06Agoda and Philippines Department of Tourism announce strategic partnership to promote and elevate tourism
[Picked up by 287 media titles]
2026-05-27 09:16Asian Society of Cardiovascular Imaging and Philips sign MOU to strengthen imaging capabilities across Asia Pacific
[Picked up by 285 media titles]
2026-05-28 17:34