Biotechnology
RemeGen's Telitacicept (RC18) Granted Fast Track Designation by United States FDA for Treatment of Primary Sjögren's Syndrome
YANTAI, China, April 3, 2024 /PRNewswire/ -- RemeGen Co. Ltd.
I-Mab Announces Closing of the Divestiture of Business Operations in China
ROCKVILLE, Md., April 2, 2024 /PRNewswire/ -- I-Mab (the "Company") (NASDAQ: IMAB) a U.S.-based, global biotech company, exclusively focused on the development and potential commercialization of highly differentiated immunotherapies for the treatment of cancer, today announced that all condition...
GC Cell to Present Multiple Posters at the American Association for Cancer Research (AACR) Annual Meeting 2024
YONGIN, South Korea, April 2, 2024 /PRNewswire/ -- GC Cell
First Patient Dosed in Clinical Trial of YOLT-101 for the Treatment of FH
SHANGHAI, April 2, 2024 /PRNewswire/ -- YolTech Therapeutics announced that the first patient has been dosed with YOLT-101, the company'sin vivo genome editing candidate being developed as a single dose, potentially curative therapy for Familial Hypercholesterolemia(FH), marking the commencement ...
Origin Agritech Provides First Half Revenue Forecast and Updates Advancements in Hybrids and GMO Development
Estimated Revenue Growth of 30-40% in the First Half of FY 2024 Two New Hybrids Completed National Trials and Four GMO Corn Hybrids Currently in National Trials BEIJING, April 2, 2024 /PRNewswire/ -- Origin Agritech Ltd. (NASDAQ: SEED) (the "Company" or "Origin"), a leading Chinese agricultural ...
CONCEPT MEDICAL RECEIVES US FDA IDE APPROVAL FOR MAGICTOUCH AVF INDICATION, THEIR FIFTH US CLINICAL STUDY APPROVAL FOR THE MAGICTOUCH PORTFOLIO
TAMPA, Fla., April 2, 2024 /PRNewswire/ -- Concept Medical has been granted 'IDE Approval' from the U.S. Food and Drug Administration (FDA) for MagicTouch AVF, its Sirolimus drug-coated balloon (DCB) catheter, to initiate a clinical study for the treatment of stenotic lesions of Arteriovenous Fis...
European Wellness Shines at AMWC 2024 in Monaco, with Prof. Dr Mike Chan and Prof. Dr Roni Moya as Faculty's Stem Cells Speakers
MONTE CARLO, Monaco, April 2, 2024 /PRNewswire/ -- The European Wellness Biomedical Group (EWBG), a global pioneer in biotechnology and bioregenerative medicine, is pleased to announce its extraordinary participation in the 22nd Aesthetic & Anti-Ageing Medicine World Congress (AMWC) 2024, which w...
Innovent and HUTCHMED Jointly Announce NDA Acceptance in China for Sintilimab Combination with Fruquintinib for the Treatment of Advanced Endometrial Cancer with Priority Review Status
ROCKVILLE, Md. and SUZHOU, China, April 2, 2024 /PRNewswire/ -- Innovent Biologics, Inc. ("Innovent") (HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures and commercializes high quality medicines for the treatment of cancer, cardiovascular and metabolic, autoimmune,...
Fosun International: Total Revenue for 2023 Reached RMB198.2 billion, Industrial Operation Profit Increased by 20% YoY
HONG KONG and SHANGHAI, April 2, 2024 /PRNewswire/ -- Fosun International Limited (HKEX stock code: 00656, "Fosun International"), together with its subsidiaries ("Fosun" or the "Group"), today announced its annual results for the year ended31 December 2023 (the "Reporting Period"). During the R...
Kemwell BioPharma Drives Quality Excellence with Veeva Vault QMS
Pioneering biologics CDMO rapidly adopts Veeva Vault QMS to advance development
and manufacturing operations
BANGALORE, India, April 2, 2024 /PRNewswire/ -- Veeva Systems
Oncoclínicas&Co has revenues of R$6 billion and profit of R$313 million in 2023
Increase in the number of patients treated and gains in operational efficiency explain record results SÃO PAULO, April 2, 2024 /PRNewswire/ -- Oncoclínicas&Co (B3: ONCO3) – the largest group dedicated to cancer treatment inLatin America – reported a 35.1% growth in its gross revenue in 2023, tot...
ThinkCyte advances biopharma phenotypic drug discovery programs with publication and presentation milestones
TOKYO, April 1, 2024 /PRNewswire/ -- ThinkCyte today announced the publication of apaper in the March issue of Cell Reports Methods on a new method for high-throughput drug discovery. Together with a joint research group including investigators at theUniversity of Tokyo and the University of Brit...
Lunit to Showcase 7 Studies at AACR 2024: Unveiling AI Innovations in HER2 Expression-Mutation Analysis and CNTN4 Biomarker Identification
- Lunit's AACR 2024 presentations to spotlight HER2 mutation insights and CNTN4's role in immunotherapy success, pioneering next-gen personalized treatment approaches, supported by the AI-powered Lunit SCOPE suite SEOUL, South Korea, April 1, 2024 /PRNewswire/ -- Lunit (KRX:328130.KQ), a leading...
Novel Immunogenicity Risk Assessment Platform, ISPRI, Facilitates Biologic Therapeutics Development
PROVIDENCE, R.I., April 1, 2024 /PRNewswire/ -- In the pursuit of developing novel biologic entities, scientists harnessing the power of AI and generative biology depend on computational tools for predicting and managing immunogenicity risk – a critical characterization step in the candidate dev...
Aculys Pharma Announces Appointment of Hidemasa Tanigaki as New CEO
~Hidemasa's appointment bolsters Aculys' position as a leader in the field of CNS inJapan and Asia~ TOKYO, April 1, 2024 /PRNewswire/ -- Aculys Pharma, Inc. ("Aculys"), a clinical-stage biopharmaceutical company focused on commercializing innovative treatments for neurological conditions, today ...
Hyosung TNC Invests $1 Billion in Future "Bio" Business
* Hyosung TNC to establish a new Bio-BDO (Butanediol) 50,000 ton factory in Vietnam with plans to expand future plants to an annual output of 200,000 tons * Replacing fossil raw materials with those from sugarcane, entering the bio raw material market * Applications across various industri...
LISCure Biosciences receives U.S. FDA Fast Track designation for LB-P8 for the treatment of primary sclerosing cholangitis (PSC)
* Phase 2 study is underway and LB-P8 is the only live biotherapeutic product currently reported to be in clinical development for the treatment of PSC * FDA's Fast Track designation for LB-P8 underlines the urgent need for treatment options to fulfill the unmet medical needs of people affecte...
73% CNS ORR! FDA Granted ODD to Utidelone Injectable (UTD1) from Biostar Pharma for the Treatment of Breast Cancer Brain Metastasis
SAN FRANCISCO, March 29, 2024 /PRNewswire/ -- Biostar Pharma, Inc., the U.S. subsidiary of Beijing Biostar Pharmaceuticals Co., Ltd. which is a synthetic biology driven biopharma company focusing on the development and commercialization of innovative oncology drugs, announced today that their cor...
Alebund's Innovative Investigational Drug AP303 Receives FDA Orphan Drug Designation (ODD) for the Treatment of Autosomal Dominant Polycystic Kidney Disease (ADPKD)
SHANGHAI, March 29, 2024 /PRNewswire/ -- Alebund Pharmaceuticals
IASO Bio Announces NMPA's IND Approval for Equecabtagene Autoleucel in Second- and Third-Line Treatment of Multiple Myeloma
SHANGHAI and NANJING, China and SAN JOSE, Calif., March 29, 2024 /PRNewswire/ -- IASO Bio, a biopharmaceutical company engaged in discovering, developing, manufacturing and marketing innovative cell therapies and antibody products, today announced thatChina National Medical Products Administratio...
Week's Top Stories
Most Reposted
Earth Day 2024: Angel Yeast Continues to Tackle Plastic Pollution Challenges With Bio-based Material Solutions
[Picked up by 294 media titles]
2024-04-22 16:00Trina Solar and PetroGreen Partner to Accelerate Philippine Solar Adoption with 117MW Supply Agreement
[Picked up by 291 media titles]
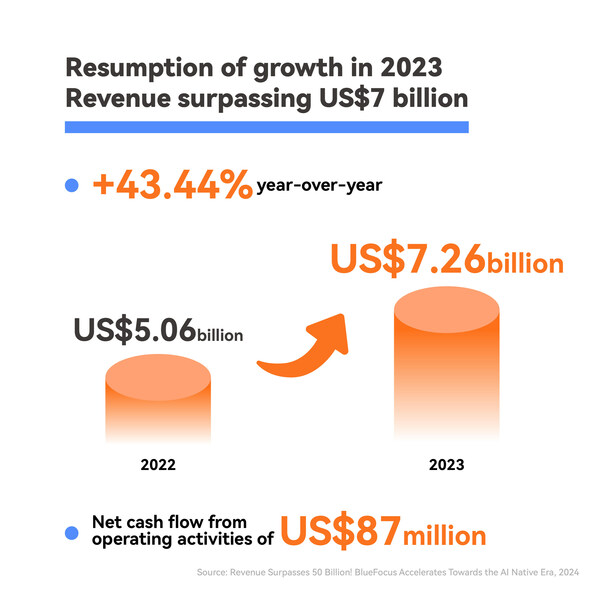
2024-04-22 06:00Revenue Surpasses 50 Billion: BlueFocus Accelerates Towards the AI Native Era
[Picked up by 275 media titles]
2024-04-23 15:43INTAMSYS Becomes 3D Printing Equipment Supplier for the WORLDSKILLS LYON 2024 COMPETITION
[Picked up by 273 media titles]
2024-04-24 17:09Introducing Wacom Movink: The first OLED pen display, and the thinnest and lightest Wacom pen display ever
[Picked up by 268 media titles]
2024-04-24 13:00