Biotechnology
PHASE II STUDY OF PAXALISIB IN BRAIN METASTASES ADVANCES TO EXPANSION STAGE IN BREAST CANCER BRAIN METASTASES COHORT
SYDNEY, June 7, 2022 Kazia Therapeutics Limited (NASDAQ: KZIA; ASX: KZA), an oncology-focused drug development company, is pleased to announce that a phase II, genomically-guided study of multiple therapies in patients with brain metastases, led by the Alliance for Clinical Trials in Oncology (NC...
Molecular Stethoscope, Inc. announces new investment from the Alzheimer's Drug Discovery Foundation
Investment by the ADDF will accelerate the translation of Molecular Stethoscope's cf-mRNA liquid biopsy technology platform into products and services for the diagnosis and management of Alzheimer's disease SOUTH SAN FRANCISCO, Calif., June 7, 2022 /PRNewswire/ -- Molecular Stethoscope, a leadin...
Chordia Therapeutics Announces Interim Results of the Phase 1 Clinical Trial of CLK Inhibitor CTX-712 at the 2022 ASCO Annual Meeting
KANAGAWA, Japan, June 7, 2022 /PRNewswire/ -- Chordia Therapeutics Inc. ("Chordia"), a biotech company engaged in the research and development of novel therapies for cancers, today announced that it has presented the interim results from the Phase 1 clinical trial of CTX-712, a selective pan-CDC-...
Chordia Therapeutics Announces Interim Results of the Phase 1 Clinical Trial of CLK Inhibitor CTX-712 at the 2022 ASCO Annual Meeting
KANAGAWA, Japan, June 7, 2022 /PRNewswire/ -- Chordia Therapeutics Inc. ("Chordia"), a biotech company engaged in the research and development of novel therapies for cancers, today announced that it has presented the interim results from the Phase 1 clinical trial of CTX-712, a selective pan-CDC-...
SCG Cell Therapy And A*STAR's IMCB Collaborate To Accelerate Clinical Translation of Immune Cell-based Therapy
* The collaboration agreement works toward accelerating the study of SCG's immunotherapy pipeline and candidates such as CAR-T, TCR-T therapies, antibodies, and vaccines. * SCG will contribute its proprietary technologies to the therapeutic development of immune cell-based therapy candidates....
Antengene Announces HREC Approval in Australia for the Phase I Trial of the Small Molecule ATR Inhibitor ATG-018
– Discovered in-house by the internal R&D Team at Antengene, ATG-018 is an orally-bioavailable, small molecule ataxia telangiectasia and Rad3-associated (ATR) kinase inhibitor that targets the DNA damage response (DDR) pathway. – This Phase I study will evaluate the safety, pharmacology and prelim...
Pharmactive Enters Strategic Partnership with Nutraconnect for APAC
Pharmactive targets Asia Pacific Market with new collaboration
MADRID, June 7, 2022 /PRNewswire/ -- Pharmactive Biotech Products
iNtRON Completes GLP-TOX Studies of BAL200
* A novel drug candidate for anthrax received orphan drug designation (ODD) by the US FDA. BOSTON and SEOUL, Korea, June 6, 2022 /PRNewswire/ -- iNtRON Biotechnology ("iNtRON" or "Company") announced today that the company has successfully completed the GLP toxicology studies of BAL200. The co...
Insilico Medicine Raises $60 Million in Series D Financing to Advance Pipeline and Launch AI-powered Drug Discovery Robotics Laboratory
NEW YORK, June 6, 2022 /PRNewswire/ -- Insilico Medicine, a clinical-stage end-to-end artificial intelligence (AI)-driven drug discovery company, announced today that it has completed a$60 million Series D financing from a syndicate of global investors with expertise in investing in the biopharm...
Senhwa's Pindnarulex in Combination Study with Pfizer's Talazoparib for the Treatment of Prostate Cancer Granted Approval to Initiate from Australian HREC
TAIPEI and SAN DIEGO, June 6, 2022 /PRNewswire/ -- Senhwa Biosciences, Inc. (TPEx: 6492), a drug development company focusing on first-in-class therapeutics for oncology, rare diseases, and novel coronaviruses, announced that it has received written approval from the Human Research Ethics Committ...
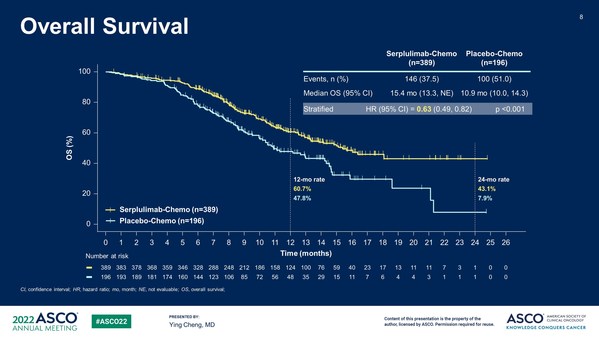
ASTRUM-005: Henlius Released Phase 3 Study Results for the First-line Treatment of Small Cell Lung Cancer of Serplulimab at ASCO 2022
SHANGHAI, June 6, 2022 /PRNewswire/ -- Shanghai Henlius Biotech, Inc. (2696.HK) announced that an international randomized phase 3 study (ASTRUM-005) of HANSIZHUANG (serplulimab), an anti-PD-1 mAb independently developed by Henlius, as first-line treatment for extensive-stage small-cell lung can...
CARsgen Therapeutics Presents Updated Data for CT041 Claudin18.2 CAR T-cells in Solid Tumors at ASCO 2022
SHANGHAI, June 6, 2022 /PRNewswire/ -- CARsgen Therapeutics Holdings Limited (Stock Code: 2171.HK), a company focused on innovative CAR T-cell therapies for the treatment of hematologic malignancies and solid tumors, announces that at the 2022 American Society of Clinical Oncology (ASCO) Annual M...
Medison Pharma Announces Multi-Regional Partnership Agreement with argenx to Commercialize Efgartigimod Across Europe and Israel
PETACH TIKVAH, Israel, June 6, 2022 /PRNewswire/ -- Medison Pharma
NeuroAiD has shown favourable outcome in patients suffering from spinal cord injuries
* SATURN study aimed at assessing the safety and effects of a 6-month course of NeuroAiD (MLC601/MLC901) to augment functional and motor recovery after severe spinal cord injury (SCI). * NeuroAiD was safe as add-on treatment in patients with severe SCI. * One third of patients improved on AS...
CPHI China opens for international partnering after China rises in CPHI Index
The CPHI China Hosted Buyer Programme is empowering new partnering in China
SHANGHAI, June 6, 2022 /PRNewswire/ -- CPHI & P-MEC China
CStone and Pfizer announce NMPA approval of sugemalimab in patients with unresectable stage III non-small cell lung cancer
* The National Medical Products Administration approved sugemalimab for the treatment of patients with unresectable stage III non-small cell lung cancer whose disease has not progressed following concurrent or sequential platinum-based chemoradiotherapy * Sugemalimab became the first anti-PD-...
CStone and Pfizer announce NMPA approval of sugemalimab in patients with unresectable stage III non-small cell lung cancer
* The National Medical Products Administration approved sugemalimab for the treatment of patients with unresectable stage III non-small cell lung cancer whose disease has not progressed following concurrent or sequential platinum-based chemoradiotherapy * Sugemalimab became the first anti-PD-...
GILEAD SCIENCES APPOINTS DUSTIN HAINES AS NEW LEADER FOR ASIA 5 REGION
-- Mr Haines Brings a Strong Track Record of Global Commercial Successes and Corporate Leadership with a Career that Spans 20 Years in Pharmaceuticals -- -- He Joins Gilead's Asia 5 Team as the Company Celebrates 10 Years of Presence inAsia -- HONG KONG, June 6, 2022 /PRNewswire/ -- Gilead Scien...
OriCiro Announces Series B2 Financing to Advance Cell-Free DNA Technology for Innovative Therapeutics and Synthetic Biology
TOKYO, June 5, 2022 /PRNewswire/ -- OriCiro Genomics, a pioneer in cell-free synthesis and amplification of genome-scale large DNA for advanced therapy and synthetic biology, today announced that it has closed Series B2 financing from Asahi Kasei Medical Co., Ltd. OriCiro has advanced its busine...
Research Updates on KN046 Presented at 2022 ASCO
SUZHOU, China, June 6, 2022 /PRNewswire/ -- Alphamab Oncology (stock code: 9966.HK) announced that research updates from four clinical studies of KN046 (PD-L1/CTLA-4 bispecific antibody) presented as a poster or published online at the 2022 American Society of Clinical Oncology Annual Meeting (20...
Week's Top Stories
Most Reposted
Earth Day 2024: Angel Yeast Continues to Tackle Plastic Pollution Challenges With Bio-based Material Solutions
[Picked up by 294 media titles]
2024-04-22 16:00Trina Solar and PetroGreen Partner to Accelerate Philippine Solar Adoption with 117MW Supply Agreement
[Picked up by 291 media titles]
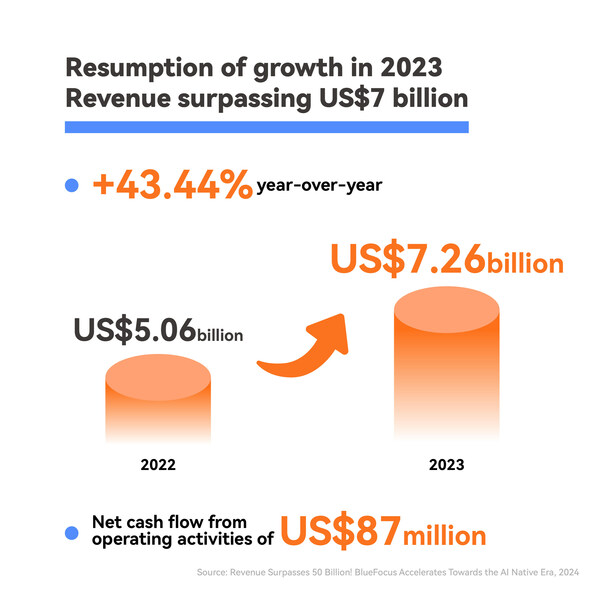
2024-04-22 06:00Revenue Surpasses 50 Billion: BlueFocus Accelerates Towards the AI Native Era
[Picked up by 275 media titles]
2024-04-23 15:43INTAMSYS Becomes 3D Printing Equipment Supplier for the WORLDSKILLS LYON 2024 COMPETITION
[Picked up by 273 media titles]
2024-04-24 17:09Introducing Wacom Movink: The first OLED pen display, and the thinnest and lightest Wacom pen display ever
[Picked up by 268 media titles]
2024-04-24 13:00