Pharmaceuticals
BioCity Announces FDA Clearance of the Investigational New Drug Application for its First-In-Class Antibody Drug Conjugate Targeting Glypican 3
SHANGHAI, April 9, 2024 /PRNewswire/ -- BioCity Biopharma today announced that the U.S. Food and Drug Administration (FDA) has cleared the company's Investigational New Drug (IND) application for a Phase 1 study of BC2027. BC2027, which is BioCity's second first-in-class antibody drug conjugate (...
YS Biopharma Announces Positive Interim Results of Pivotal Phase 3 Clinical Study of PIKA Rabies Vaccine
GAITHERSBURG, Md., April 9, 2024 /PRNewswire/ -- YS Biopharma Co., Ltd. (Nasdaq: YS) ("YS Biopharma" or the "Company"), a global biopharmaceutical company dedicated to discovering, developing, manufacturing, and delivering new generations of vaccines and therapeutic biologics for infectious disea...
Jacobio Pharma to Present Data of PARP7 Inhibitor and P53 Reactivator at the 2024 AACR Annual Meeting
BEIJING, SHANGHAI, and SAN DIEGO, April 9, 2024 /PRNewswire/ -- Jacobin Pharma (1167.HK), a clinical-stage oncology company focusing on undruggable targets, today announced that the company will present the results of two preclinical evaluation of PARP7 inhibitor JAB-26766 and p53 Y220C reactivat...
GenFleet Therapeutics Announces Broad-spectrum Activity of GFH547, an Oral panRAS (ON) Inhibitor, and Its Potential to Overcome Resistance against SIIP-based KRAS Inhibitors in Late-breaking Research Abstract of 2024 AACR Annual Meeting
SHANGHAI and SAN DIEGO, April 9, 2024 /PRNewswire/ -- GenFleet Therapeutics, a clinical-stage biotechnology company focusing on cutting-edge therapies in oncology and immunology, today announced the latest findings of GFH547, an oral panRAS (ON) inhibitor, in a late-breaking research abstract at ...
GenFleet Therapeutics Announces Potent Anti-tumor Efficacy of GFH375, an Oral KRAS G12D (ON/OFF) Inhibitor, and its Potential in Combination Therapy with RAF/MEK Clamp at 2024 AACR Annual Meeting
SHANGHAI and SAN DIEGO, April 9, 2024 /PRNewswire/ -- GenFleet Therapeutics, a clinical-stage biotechnology company focusing on cutting-edge therapies in oncology and immunology, today unveiled the latest research findings of GFH375, an oral KRAS G12D (ON/OFF) inhibitor, at the poster presentatio...
Fiscal Year 2023: Exyte experiences strong growth in Europe and the USA
* Sales double in the USA, order intake increases by over 150 percent * More than 50 percent increase in order intake in EMEA * Business Area Technology & Services' sales grow by almost 25 percent * Further improvement in profitability * Exyte CEO Büchele: "Our follow-the-client strategy ...
Waters Launches Alliance iS Bio HPLC System to Help Biopharma QC Labs Increase Efficiency and Reduce Errors
News Summary: * New Alliance™ iS Bio High Performance Liquid Chromatography (HPLC) System helps biopharmaceutical QC laboratories boost efficiency and eliminate up to 40% of common errorsi. * Featuring MaxPeak™ High Performance Surface (HPS) Technology, the new system minimizes unwanted anal...
Transcenta Announces Collaboration with Agilent to Develop a Claudin18.2 Companion Diagnostic to Support Osemitamab (TST001) Global Phase III Trial
PRINCETON, N.J. and SUZHOU, China, April 9, 2024 /PRNewswire/ -- Transcenta Holding Limited ("Transcenta") (HKEX: 06628), a clinical stage biopharmaceutical company with fully-integrated capabilities in discovery, research, development and manufacturing of antibody-based therapeutics, and Agilen...
Oral report | The positive interim analysis results from the Phase III clinical study of Akeso's Cadonilimab (PD-1/CTLA-4 BsAb) plus chemotherapy as first-line treatment for advanced gastric cancer presented at 2024 AACR
SAN DIEGO, April 8, 2024 /PRNewswire/ -- Akeso, Inc. (9926.HK) today announced thatthe positive interim analysis results from Phase III clinical study of Cadonilimab (PD-1/CTLA-4 BsAb)combined with XELOX (capecitabine plus oxaliplatin) as first-line treatment for unresectable locally advanced or ...
MAIWEIJIAN, First Approved Biosimilar of Denosumab (120mg) in China
SHANGHAI, April 8, 2024 /PRNewswire/ -- Mabwell (688062.SH), an innovation-driven biopharmaceutical company with entire industry chain, announced that Denosumab Injection (trade name: MAIWEIJIAN, R&D code: 9MW0321) developed by its wholly-owned subsidiary T-mab has officially obtained the market...
SeekIn Presents New OncoSeek® Data at the AACR Annual Meeting in San Diego
Two Sample Types, Three Different Platforms, Six Independent Cohorts of ~12,000 Cancer Patients and Non-cancer Individuals with Diverse Ethnic Backgrounds in Retrospective and Prospective Settings Validate OncoSeek's Ability to Detect Multiple Cancer Types and Predict the Tissue of Origin of Ca...
Seegene and UK Health Security Agency convene a summit to collectively realize "a World Free from All Diseases"
* Two CEOs met at Seegene headquarters to discuss and concurred with Seegene's vision * Seegene's swift responses to the COVID-19 pandemic and digital solutions through AI implementation were presented SEOUL, South Korea, April 8, 2024 /PRNewswire/ -- Seegene Inc., a leading South Korean comp...
Clover Announces Positive Preliminary Phase I Results for Bivalent RSV Vaccine Candidate SCB-1019 in Initial Young Adult Cohort
-- Bivalent SCB-1019 significantly boosted RSV-A and RSV-B neutralization titers to approximately 6,600 IU/mL (6.4-fold increase) and approximately 46,000 IU/mL (12-fold increase), respectively -- -- Preliminary Phase I data in target older adult population is on track for H2-2024 -- SHANGHAI, ...
D3 Bio Completes Series A+ Round to Advance Innovative Oncology Pipeline
SHANGHAI, April 8, 2024 /PRNewswire/ -- D3 Bio, an emerging global biotechnology company that focuses on discovery, development, and registration of innovative cancer drugs announces its closing of Series A+ financing round led by Medicxi, a leading European life sciences investment firm. D3 Bio'...
Laekna Announces Two Poster Presentations on Internally Discovered Drug Candidates at AACR 2024
SHANGHAI and WARREN, N.J., April 7, 2024 /PRNewswire/ -- Laekna (2105.HK) announced that the company has presented two internally-discovered preclinical candidates, in addition to a poster presentation on a clinical trial, at the 2024 Annual Meeting of the American Association for Cancer Research...
Innovent to Present Preclinical Data of Multiple Novel Molecules at the 2024 AACR Annual Meeting
ROCKVILLE, Md. and SUZHOU, China, April 8, 2024 /PRNewswire/ -- Innovent Biologics, Inc. ("Innovent") (HKEX: 01801), a world-class biopharmaceutical company that develops, manufactures and commercializes high quality medicines for the treatment of oncology, cardiovascular and metabolic, autoimmun...
FDA Grants Breakthrough Therapy Designation to Sunvozertinib for the First-Line Treatment of Patients with advanced Non-Small Cell Lung Cancer Harboring EGFR Exon 20 Insertion Mutations
SHANGHAI, April 7, 2024 /PRNewswire/ -- Dizal (688192.SH) today announced that the U.S. Food and Drug Administration ("FDA") has granted Breakthrough Therapy Designation (BTD) to its sunvozertinib as the first-line treatment for patients with locally advanced or metastatic non-small cell lung can...
Antengene Presents Four Preclinical Posters at AACR 2024
SHANGHAI and HONG KONG, April 6, 2024 /PRNewswire/ -- Antengene Corporation Limited ("Antengene", SEHK: 6996.HK), a leading innovative, commercial-stage global biopharmaceutical company dedicated to discovering, developing and commercializing first-in-class and/or best-in-class medicines for canc...
I-Mab to Participate at the 23rd Annual Needham Virtual Healthcare Conference
ROCKVILLE, Md., April 5, 2024 /PRNewswire/ -- I-Mab (NASDAQ: IMAB), a U.S.-based, global biotech company, exclusively focused on the development and potential commercialization of highly differentiated immunotherapies for the treatment of cancer, today announced that management will present at th...
HSA approves new asthma indication for GSK's Trelegy Ellipta, the first once-daily single inhaler triple therapy approved for use in the treatment of both asthma and COPD in Singapore
SINGAPORE, April 5, 2024 /PRNewswire/ -- GSK Singapore announced that the Health Sciences Authority (HSA) has approved a new indication and a new strength for Trelegy Ellipta (fluticasone furoate/umeclidinium/vilanterol 'FF/UMEC/VI') for the maintenance treatment of asthma in patients aged 18 yea...
Week's Top Stories
Most Reposted
Earth Day 2024: Angel Yeast Continues to Tackle Plastic Pollution Challenges With Bio-based Material Solutions
[Picked up by 294 media titles]
2024-04-22 16:00Trina Solar and PetroGreen Partner to Accelerate Philippine Solar Adoption with 117MW Supply Agreement
[Picked up by 291 media titles]
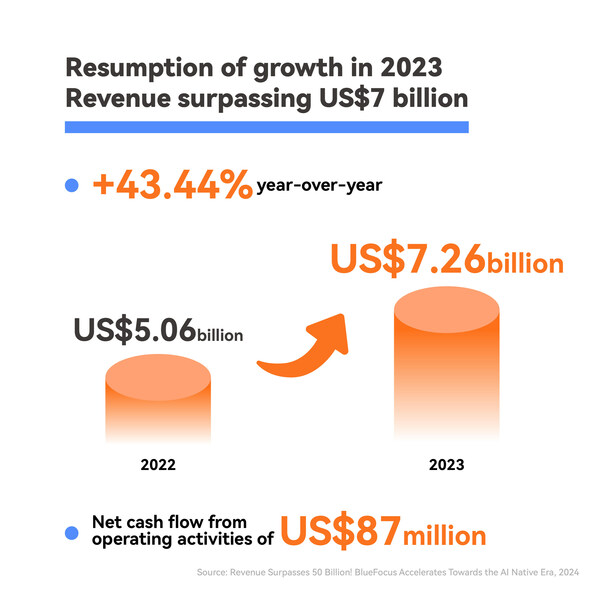
2024-04-22 06:00Revenue Surpasses 50 Billion: BlueFocus Accelerates Towards the AI Native Era
[Picked up by 275 media titles]
2024-04-23 15:43INTAMSYS Becomes 3D Printing Equipment Supplier for the WORLDSKILLS LYON 2024 COMPETITION
[Picked up by 273 media titles]
2024-04-24 17:09Introducing Wacom Movink: The first OLED pen display, and the thinnest and lightest Wacom pen display ever
[Picked up by 268 media titles]
2024-04-24 13:00