Harbour BioMed Announces First Subject Dosed in Phase I Study of Next-Gen Anti-TSLP Fully Human Monoclonal Antibody
CAMBRIDGE, Mass., ROTTERDAM, Netherlands and SUZHOU, China, Sept. 27, 2022 /PRNewswire/ -- Harbour BioMed ("HBM", HKEX: 02142) today announces that it has completed the first subject dosing in a phase I study of HBM9378 (or SKB378 as referred to by Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd...
Harbour BioMed to Announce 2022 Interim Results on August 31, 2022
CAMBRIDGE, Mass., ROTTERDAM, Netherlands and SUZHOU, China, Aug. 9, 2022 /PRNewswire/ -- Harbour BioMed (the "Company", HKEX: 02142) announced that it will report financial results for the first half year endedJune 30, 2022, on Wednesday, August 31, 2022. The Company will host an English virtual ...
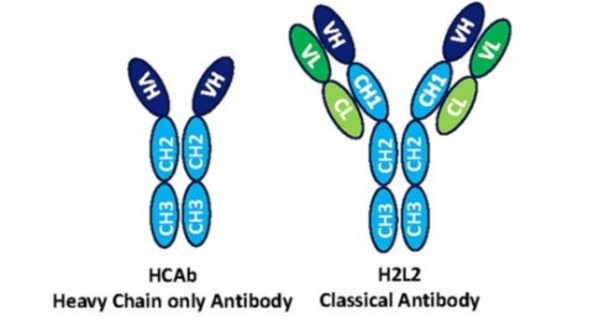
PNAS Published Preclinical Results of Harbour BioMed's Next-Generation Fully Human Heavy-chain Antibody Porustobart
CAMBRIDGE, Mass., ROTTERDAM, Netherlands and SUZHOU, China, Aug. 8, 2022 /PRNewswire/ -- Harbour BioMed (the "Company", HKEX: 02142) announced that the preclinical results of porustobart (HBM4003,or the HCAb 4003-2 in the research paper), a next-generation fully human heavy-chain antibody with a ...
Harbour BioMed Announces IND Clearance for B7H4x4-1BB Bispecific Antibody by the U.S. Food and Drug Administration
CAMBRIDGE, Mass., ROTTERDAM, Netherlands and SUZHOU, China, June 27, 2022 /PRNewswire/ -- Harbour BioMed ("HBM", HKEX: 02142) announced that the U.S. Food and Drug Administration (FDA) has cleared the Investigational New Drug (IND) application for the B7H4x4-1BB bispecific antibody (HBM7008), whi...
Harbour BioMed Announces IND Approval for B7H4x4-1BB Bispecific Antibody
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, June 8, 2022 /PRNewswire/ --Harbour BioMed ("HBM", HKEX: 02142) announced that China National Medical Products Administration (NMPA) had approved the investigational new drug (IND) application to commence phase I trial of its B7H4x4-...
Harbour BioMed Reports the Latest Progress of Next-Generation Fully Human Heavy-chain Antibody HBM4003 with Unique Treg Depletion Mechanism
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, June 1, 2022 /PRNewswire/ -- Harbour BioMed (the "Company", HKEX: 02142), a global biopharmaceutical company committed to the discovery, development, and commercialization of novel antibody therapeutics, today announced the progress ...
Harbour BioMed Announces Dosing of First Patient in Phase I Trial of B7H4x4-1BB Bispecific Antibody
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, May 25, 2022 /PRNewswire/ -- Harbour BioMed ("HBM", HKEX: 02142) announcedthat it has successfully completed the dosing of first patient in phase I trial of B7H4x4-1BB bispecific antibody HBM7008 inAustralia. This study will evaluate ...
Harbour BioMed to Present the Latest Progress of Next-Generation Anti-CTLA-4 Antibody HBM4003 at 2022 ASCO Annual Meeting
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, May 10, 2022 /PRNewswire/ -- Harbour BioMed (the "Company", HKEX: 02142), a global biopharmaceutical company committed to the discovery, development, and commercialization of novel antibody therapeutics, today announced the Company w...
Harbour BioMed Appoints Dr. Alexander A. Zukiwski to its Scientific Advisory Board
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, May 5, 2022 /PRNewswire/ -- Harbour BioMed ("HBM", HKEX:02142), a global biopharmaceutical company committed to the discovery, development, and commercialization of novel antibody therapeutics, is pleased to announce the appointment o...
Harbour BioMed Appoints Dr. Humphrey Gardner as Chief Medical Officer
CAMBRIDGE, Mass., SUZHOU, China and ROTTERDAM, Netherlands, April 13, 2022 /PRNewswire/ -- Harbour BioMed ("HBM" or the "Company"; HKEX: 02142), today announced the appointment of Dr.Humphrey Gardner as Chief Medical Officer (CMO). Dr. Gardner will be responsible for leading the global product d...
Harbour BioMed Announces Global Out-License Agreement with AstraZeneca for CLDN18.2xCD3 Bispecific Antibody HBM7022
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, April 6, 2022 /PRNewswire/ -- Harbour BioMed ("HBM", "the Company", HKEX: 02142), a global biopharmaceutical company committed to the discovery, development, and commercialization of novel antibody therapeutics, today announced a glob...
Harbour BioMed Announces Poster Presentation on the Novel Bispecific Antibody PD-L1xCD40 at Upcoming American Association for Cancer Research (AACR) Annual Meeting
CAMBRIDGE, Mass., ROTTERDAM, Netherlands and SUZHOU, China, April 6, 2022 /PRNewswire/ -- Harbour BioMed ("HBM", HKEX: 02142), a global biopharmaceutical company committed to the discovery, development, and commercialization of novel antibody therapeutics, today announced that the poster presenta...
Harbour BioMed Reports Full Year 2021 Financial Results: Empower Value Creation with Global Innovation Capabilities
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, March 24, 2022 /PRNewswire/ --Harbour BioMed ("HBM" or the "Company"; HKEX: 02142), a global biopharmaceutical company committed to the discovery, development and commercialization of novel antibody therapeutics focusing on oncology a...
Harbour BioMed Announces IND Approval for Next-Gen Anti-TSLP Fully Human Monoclonal Antibody
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, Feb. 21, 2022 /PRNewswire/ -- Harbour BioMed ("HBM", HKEX: 02142) announced that China National Medical Products Administration (NMPA) had approved its investigational new drug (IND) application for HBM9378 (or SKB378 as referred to ...
Harbour BioMed Announces Dosing of First Patient in Combination Therapy Phase Ib/IIa Trial of Next-Generation Anti-CTLA-4 Antibody
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, Jan. 24, 2022 /PRNewswire/ -- Harbour BioMed ("HBM", HKEX: 02142) announced that, it has successfully completed the dosing of first patient in phase Ib/IIa trial at the stage of dose expansion of its anti-CTLA-4 antibody (HBM4003) in ...
Harbour BioMed Announces Dosing of First Patient of Two Phase I Trials for Next-Generation Anti-CTLA-4 Antibody HBM4003
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, Jan. 4, 2022 /PRNewswire/ -- Harbour BioMed ("HBM", HKEX: 02142) announced that, its next generation anti-CTLA-4 fully human heavy-chain only antibody (HCAb), HBM4003, has completed the first dosing of first patient in two phase I tri...
Harbour BioMed Announces Dosing of First Patient of Batoclimab Phase II Trial in Patients with Thyroid Eye Disease
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, Oct. 19, 2021 /PRNewswire/ -- Harbour BioMed ("HBM", HKEX: 02142) announced that, the clinical trial of its anti-FcRn antibody, batoclimab (HBM9161), has completed the first dosing of first patient in phase II trial for the treatment ...
Harbour BioMed Announces Dosing of First Patient of Batoclimab Phase III Trial in Patients with Generalized Myasthenia Gravis
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, Sept. 27, 2021 /PRNewswire/ -- Harbour BioMed ("HBM", HKEX: 02142) announced that, the clinical trial of its anti-FcRn antibody, batoclimab (HBM9161), has completed the first dosing of first patient in registrational phase III trial f...
Harbour BioMed Announces Phase I Study Positive Results on Next-Generation Anti-CTLA-4 Antibody HBM4003
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, Sept. 13, 2021 /PRNewswire/ -- Harbour BioMed ("HBM", HKEX: 02142) announced positive results from its phase I dose escalation clinical trial of HBM4003 in solid tumors in Australia (the "phase I study"). The clinical data abstract has...
Harbour BioMed Announces IND Approval for Phase II Trial of Anti-FcRn Batoclimab in Chronic Inflammatory Demyelinating Polyneuropathy in China
CAMBRIDGE, Mass. and ROTTERDAM, Netherlands and SUZHOU, China, Sept. 1, 2021 /PRNewswire/ -- Harbour BioMed ("HBM", HKEX: 02142) today announced that the Center for Drug Evaluation (CDE) of China National Medical Products Administration (NMPA) has approved the Investigational New Drug (IND) appl...