Medical/Pharmaceuticals
World Lupus Federation Global Survey Finds 91% of People with Lupus Report Using Oral Steroids to Treat Lupus
Global public urged to share survey results and lupus facts on May 10 for World Lupus Day WASHINGTON, May 6, 2024 /PRNewswire/ -- A recent global survey conducted by the World Lupus Federation reported that 91% of survey respondents were taking or have taken oral steroids to treat their lupus. O...
VYNDAMAX® (tafamidis) PBS-listed for adult patients with wild-type or hereditary transthyretin amyloid cardiomyopathy (ATTR-CM) with New York Heart Association (NYHA) Class I-II heart failure
* VYNDAMAX has been listed on the Pharmaceutical Benefits Scheme (PBS) for adult patients with wild-type or hereditary transthyretin amyloid cardiomyopathy (ATTR-CM)with New York Heart Association (NYHA) Class I-II heart failure.[1] * ATTR-CM is a debilitating and often fatal condition that l...
United Imaging to Unveil Industry-Leading MRI Advancements at ISMRM 2024
Ultra-high-field technology, ultra-high-gradient technology, and advances in artificial intelligence take center stage. SHANGHAI, May 4, 2024 /PRNewswire/ -- United Imaging, a global leader in manufacturing advanced medical imaging and radiotherapy equipment, will unveil its latest MRI technolog...
HanAll Biopharma Announces Initiation of Phase III Randomized, Double-Masked Vehicle Controlled VELOS-4 Trial Evaluating Tanfanercept for Treatment of Dry Eye Disease
* HanAll has initiated a Phase III VELOS-4 study to evaluate the efficacy and safety of tanfanercept in dry eye based on the findings from the previous Phase III VELOS-3 study. * Tanfanercept demonstrated statistically significant improvement on the secondary outcome measure, Schirmer testing...
Clarivate Enhances Cortellis CMC Intelligence with Post-Approval Module to Accelerate Regulatory Success
Enhancements enable pharma, biotech and generics companies to streamline regulatory tracking and optimize life cycle management for small molecules and biologics LONDON, May 2, 2024 /PRNewswire/ -- Clarivate Plc (NYSE:CLVT), a leading global provider of transformative intelligence, today announ...
Vivo Surgical Announces Leading Gastrointestinal Endoscopists on Clinical Advisory Board for Novel Endoscopic Robot
Inaugural international clinical advisory board to provide strategic guidance towards development of the company's ERES™ endoscopic robot SINGAPORE, May 2, 2024 /PRNewswire/ -- Vivo Surgical Private Limited, a privately-held surgical technology company pioneering a flexible endoscopic robot for ...
Everest Medicines Announces Hong Kong Department of Health Approval of Nefecon® for the Treatment for Primary IgA Nephropathy in Adult Patients
SHANGHAI, May 2, 2024 /PRNewswire/ -- Everest Medicines (HKEX 1952.HK, "Everest", or the "Company") announced that the Hong Kong Department of Health had approved Nefecon® for the treatment of primary immunoglobulin A nephropathy (IgAN) in adults at risk of disease progression.Hong Kong marks the...
Integrated cancer biotech Infinitopes secures £12.8m seed financing to enhance its Precision Immunomics™ antigen discovery technologies to target five more cancers
Funding also supports the start of phase I/IIa study for lead vaccine, ITOP1 OXFORD, England, May 2, 2024 /PRNewswire/ -- Infinitopes Precision Immunomics, an integrated cancer biotech combining world leading platforms in precision antigen discovery with vaccine vectors capable of durably stimula...
United Imaging Receives FDA Clearance for Breakthrough 5T MRI System
As the company heads to ISMRM, the world's first whole-body Ultra-High Field MRI officially comes to market. HOUSTON, May 2, 2024 /PRNewswire/ -- United Imaging, a global leader in manufacturing advanced medical imaging and radiotherapy equipment, has achieved a significant milestone with the U....
3Shape Unveils Unite 3rd Generation: Access TRIOS Scans Anywhere, Anytime
COPENHAGEN, Denmark, May 1, 2024 /PRNewswire/ -- 3Shape launches Unite 3rd Generation, allowing dental professionals to access and work with their digital impressions and patient cases anywhere, anytime, via any mobile, tablet (Android, iOS), or web device. This update enables seamless and secure...
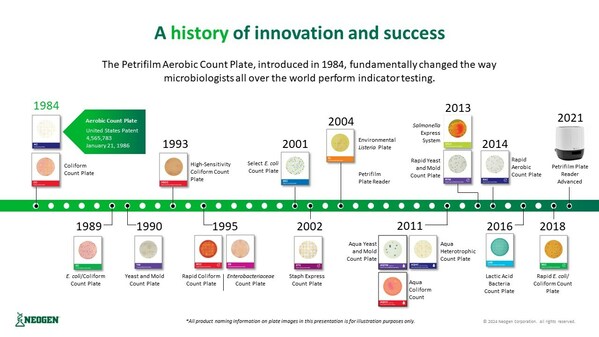
Neogen® Petrifilm® Celebrates 40 Years!
LANSING, Mich., May 1, 2024 /PRNewswire/ -- Neogen® Corporation (NASDAQ: NEOG) is celebrating 40 years of Petrifilm, the innovation that changed how microbiologists around the world perform indicator testing. "In the food safety industry, Petrifilm is recognized as a true game-changer," saidJohn...
ArisGlobal Helps Boehringer Ingelheim Transform Safety Signal Processing by Leveraging Latest LifeSphere Solutions
LifeSphere Clarity drives efficiencies and reinvents the way Boehringer Ingelheim's pharmaceutical operations approach safety signals BOSTON, May 1, 2024 /PRNewswire/ -- ArisGlobal, a market leader in Life Sciences technology and the creator of LifeSphere®, announced today that research-driven g...
135th Canton Fair Showcases Leading Medical Devices, Elevating Global Health Management
GUANGZHOU, China, May 1, 2024 /PRNewswire/ -- In a significant move to meet the growing demand for health management solutions, the 135th China Import and Export Fair ("Canton Fair" or "the Fair") has brought together top-tier companies from theMedicines, Health Products and Medical Devices secto...
Jacobio Pharma Announced its KRAS G12C inhibitor reached the primary endpoint
BEIJING and SHANGHAI and BOSTON, April 30, 2024 /PRNewswire/ -- Jacobio Pharma (1167.HK) announced that the data from the Phase II registrational study of the KRAS G12C inhibitor glecirasib were offically reported at the April ASCO Plenary Series, which was held online. Prof. Yuankai Shi, chief ...
Integrated cancer biotech Infinitopes secures £12.8m seed financing to enhance its Precision Immunomics™ antigen discovery technologies to target five more cancers
Funding also supports the start of phase I/IIa study for lead vaccine, ITOP1 OXFORD, England, May 1, 2024 /PRNewswire/ -- Infinitopes Precision Immunomics, an integrated cancer biotech combining world leading platforms in precision antigen discovery with vaccine vectors capable of durably stimula...
Eluminex Biosciences Announces FDA Acceptance of Investigational New Drug (IND) Application for EB-105 - A Novel Trispecific Fusion Antibody for Diabetic Macular Edema (DME) - and Upcoming Scientific Presentations
SAN FRANCISCO and SUZHOU, China, April 30, 2024 /PRNewswire/ -- Eluminex Biosciences (Eluminex), a privately-held biotechnology company focused on the development of advanced protein therapeutics for vision-threatening diseases and dermal facial aesthetics announced the acceptance of their EB-105...
National Medical Products Administration (NMPA) Approves Chipscreen Bioscience's Chidamide (Epidaza) combined with R-CHOP for the treatment of diffuse large B-cell lymphoma
SHENZHEN, China, April 30, 2024 /PRNewswire/ -- Shenzhen Chipscreen Biosciences Co., Ltd. (Chipscreen Biosciences, Stock Symbol: 688321.SH) announced that the company's lead innovative product Chidamide (Epidaza®) , an oral subtype-selective histone deacetylase (HDAC) inhibitor, combined with R-C...
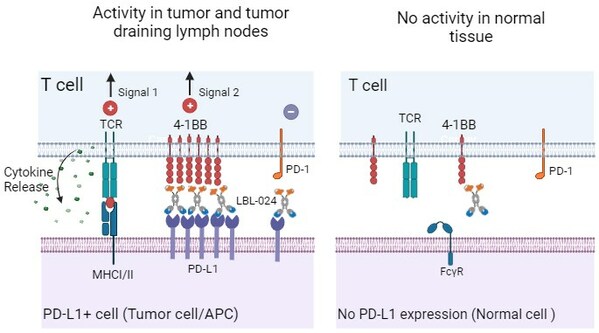
A Potential First-In-Class Drug: CDE Approved Single-Arm Pivotal Clinical Study of LBL-024, An Anti-PD-L1/4-1BB Bispecific Antibody Developed by Leads Biolabs
NANJING, China, April 30, 2024 /PRNewswire/ -- Nanjing Leads Biolabs Co., Ltd. (hereinafter referred to as "Leads Biolabs") announced that LBL-024, an anti-PD-L1/4-1BB bispecific antibody independently developed by Leads Biolabs with global intellectual property rights has received approval to co...
Medison Pharma and Alnylam Pharmaceuticals Announce Expansion of their Multi-Regional Partnership in Europe and Israel to Commercialize RNAi Therapeutics in additional LATAM and APAC markets including Australia
* The expanded partnership will allow Alnylam and Medison to help accelerate access for patients in multiple regions under one global alliance * Medison, the creator and leader of the multi-regional partnership category, will utilize its unique, unifiedplatform for efficient global commerciali...
HanAll Biopharma Reports Q1 2024 Financial Results and Provides Business Update
* Delivered solid performance to start 2024, with record-breaking first quarter revenue of34.1 billion KRW. Strong sales momentum continued from key products, funding investments in ongoing R&D programs. * Phase 3 VELOS-4 study of tanfanercept in dry eye disease expected to be initiated in th...
Week's Top Stories
Most Reposted
Tetra Pak Opens Product Development Centre in Thailand, Unlocking A World of Possibilities for Food and Beverage Innovators Across Asia Pacific
[Picked up by 316 media titles]
2026-03-27 11:15Visa Working Capital Index: Asia Pacific CFOs Call for Flexible, Digital Finance Solutions
[Picked up by 310 media titles]
2026-03-24 14:00Indonesia's P2P Lending Grows 25%, JULO Strengthens Credit Quality
[Picked up by 300 media titles]
2026-03-26 18:18ART CENTRAL COMMENCES DISCOVERY-LED ELEVENTH EDITION
[Picked up by 279 media titles]
2026-03-25 16:51Noah Reports Q4 and Full Year 2025 Earnings: Structural Transformation Driving Significant Improvement in Profitability as AI Integration Drives Operational Efficiency
[Picked up by 275 media titles]
2026-03-25 21:58