Pharmaceuticals
Veltassa® (patiromer) approved in Japan for the treatment of adults with hyperkalemia
Veltassa® offers effective and well-tolerated long-term potassium control in chronic kidney disease and chronic heart failure patients1-5 Zeria Pharmaceutical Co., Ltd. to market Veltassa® in Japan ST. GALLEN, Switzerland, Sept. 24, 2024 /PRNewswire/ -- CSL Vifor today announced thatJapan's Mini...
Antengene Announces XPOVIO® (selinexor) Approved for Commercialization in Thailand
* XPOVIO® is the first and only approved XPO1 inhibitor in Thailand. * XPOVIO® has been approved for multiple indications in nine markets across the APAC region. Antengene has submitted a new drug application (NDA) for XPOVIO ® in Indonesia with approval expected in the second half of 2024. ...
Impressive preliminary objective response rates (ORR) of 36% in NSCLC and 80% in EGFRmut NSCLC: Interim Results of a Phase 1 Study of BC3195, a First-in-Class ADC Targeting CDH3, Presented by BioCity at ESMO 2024
SHANGHAI, Sept. 23, 2024 /PRNewswire/ -- On September 14, 2024, BioCity presented interim clinical results on the safety and efficacy of its first-in-class antibody-drug conjugate (ADC) BC3195 targeting CDH3 (P-Cadherin) in a Phase I clinical trial at the European Society for Medical Oncology (ES...
WuXi Biologics Included in Hang Seng ESG 50 Index
HONG KONG, Sept. 23, 2024 /PRNewswire/ -- WuXi Biologics ("WuXi Bio") (2269.HK), a leading global Contract Research, Development and Manufacturing Organization (CRDMO), today announced that it has been included in the Hang Seng ESG 50 Index, making it the only pharmaceutical company selected this...
Professor Luo Min Appointed as Chairman of Spine Neurocardiology Interdisciplinary Professional Committee in China to Promote Development of Innovative Heart Disease Treatments
BEIJING, Sept. 23, 2024 /PRNewswire/ -- Professor Luo Min, Doctor of Medicine (MD), from Beijing Yuhe Integrated Traditional Chinese and Western Medicine Rehabilitation Hospital, was recently appointed as the Chairman of the National Association of Health Industry and Enterprise Management (NAHIE...
Nuance Pharma Announces Expansion of Bentrio License and Distribution Agreement with Altamira Therapeutics in East and South East Asia
* Nuance Pharma successfully launched Bentrio® nasal spray for protection against airborne particles inHong Kong and filed request for marketing authorization for Mainland China * Extension of Exclusive License and Distribution Agreement to include seven additional countries across East andS...
Telix to Acquire RLS to Expand North American Manufacturing and Distribution Platform
MELBOURNE, Australia, INDIANAPOLIS and LAKE MARY, Fla., Sept. 23, 2024 /PRNewswire/ -- Telix Pharmaceuticals Limited (ASX: TLX, Telix, the Company) and RLS (USA) Inc. (RLS, RLS Radiopharmacies), America's only Joint Commission-accredited radiopharmacy network distributing PET[1], SPECT[2] and th...
Henlius Receives Positive CHMP Opinion for HANSIZHUANG as First-Line Treatment for Extensive-Stage Small Cell Lung Cancer
* HANSIZHUANG (serplulimab) is the world's first anti-PD-1 mAb for first-line treatment of ES- SCLC - * HANSIZHUANG (serplulimab) is expected to become the first and only anti-PD-1 monoclonal antibody available inEurope for first-line treatment of ES- SCLC - * HANSIZHUANG (serplulimab) ha...
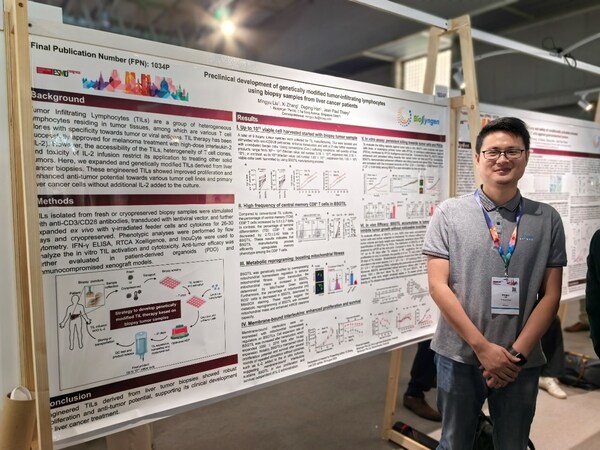
Biosyngen Best-in-Class Next-Generation Tumor-Infiltrating Lymphocyte (TIL) Technology Debuts on ESMO 2024 Annual Meeting
BARCELONA, Spain, Sept. 20, 2024 /PRNewswire/ -- The highly anticipated 2024 European Society for Medical Oncology (ESMO) Annual Congress has taken place fromSeptember 13 to 17 in Barcelona, Spain. As one of the most influential annual gatherings in oncology, this congress brings together leading...
Kangpu Completed Bridging Clinical Study of KPG-818 in China
HEFEI, China, Sept. 20, 2024 /PRNewswire/ -- Kangpu Biopharmaceuticals, a clinical-stage company based inHefei, China, announced today that the Company has successfully completed a bridging clinical study of KPG-818 in healthy subjects inChina. The randomized, double-blind, placebo-controlled,...
CD (Suzhou) Biopharma Announces FDA Clearance for Phase I Clinical Trial of CD-001
SUZHOU, China, Sept. 20, 2024 /PRNewswire/ -- CD (Suzhou) Biopharma
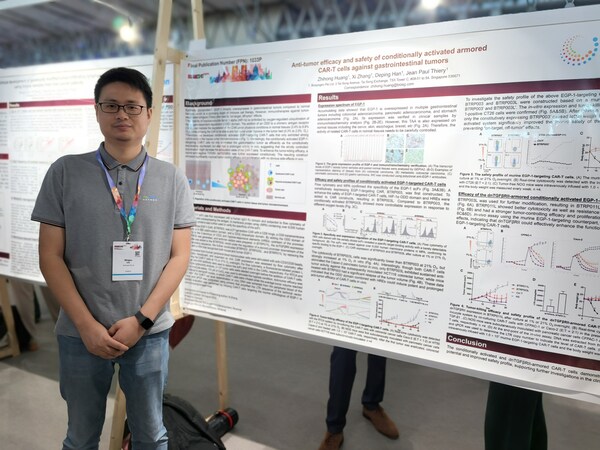
Biosyngen Presents Pioneering"Conditional Activation + Armor Enhancement" SUPER-T technology at ESMO 2024
BARCELONA, Spain, Sept. 19, 2024 /PRNewswire/ -- The highly anticipated 2024 European Society for Medical Oncology (ESMO) Annual Congress has taken place fromSeptember 13 to 17 in Barcelona, Spain. As one of the most influential annual gatherings in oncology, this congress brings together leading...
Cambrex Announces New Liquid-Phase Peptide Synthesis Manufacturing Technology
EAST RUTHERFORD, N.J., Sept. 19, 2024 /PRNewswire/ -- Cambrex, a leading global contract development and manufacturing organization (CDMO), today announced that Snapdragon Chemistry, a Cambrex company, has successfully developed a new liquid-phase peptide synthesis (LPPS) technology that utilizes...
Sirnaomics Announces Demonstration of a Novel Mechanism of Action of Its siRNA Therapeutics for Focal Fat Reduction in Journal of Cosmetic Dermatology
HONG KONG, GERMANTOWN, Md., and SUZHOU, China, Sept. 19, 2024 /PRNewswire/ -- Sirnaomics Ltd.(the "Company", Stock Code: 2257, together with its subsidiaries, the "Group" or "Sirnaomics"), a leading biopharmaceutical company engaging in discovery and development of advanced RNAi therapeutics, tod...
Hyphens Pharma Launches Ceradan® Advanced Hand Balm, Specially Designed for Hand Eczema
SINGAPORE, Sept. 19, 2024 /PRNewswire/ -- Today, Hyphens Pharma International Limited ("Hyphens Pharma", or the "Company", and together with its subsidiaries, the "Group"), Singapore's leading specialty pharmaceutical and consumer healthcare group, is pleased to announce the launch of Ceradan® A...
Best Poster of ESMO 2024! Abbisko Announces Updated Clinical Data of Irpagratinib in HCC
SHANGHAI, Sept. 18, 2024 /PRNewswire/ -- Abbisko Therapeutics (HKEX: 02256) is excited to announce the receipt of the ESMO 2024 Best Poster Award on September 16, 2024. The award was received for the presentation titled "Updated Safety and Efficacy of Irpagratinib (ABSK011) in advanced hepatocell...
Transcenta Updates Encouraging Efficacy Data from First-line Triple Combo Trial of Osemitamab (TST001) for G/GEJ Cancer at ESMO 2024
Updated results with longer follow-up reveal confirmed ORR of 68% and median PFS of 14.2 months in patients with CLDN18.2 high or medium expression, known PD-L1 CPS (n=66). PRINCETON, N.J. and SUZHOU, China, Sept. 19, 2024 /PRNewswire/ -- Transcenta Holding Limited ("Transcenta") (HKEX: 06628), ...
Insilico Medicine Reports Positive Phase IIa Results for ISM001-055, a Novel First-in-Class Drug Treatment for Idiopathic Pulmonary Fibrosis (IPF) Designed Using Generative AI
* ISM001-055 is a novel drug designed in-house using generative AI to target TNIK (Traf2- and NCK- interacting kinase) and has progressed through Phase IIa clinical testing. * Preliminary results from this 12-week study demonstrated that ISM001-055 possesses a favorable safety profile and dos...
Bloomage 2024 Mid-Year Report: Driving Growth Through Strategic Transformation to a Solutions Provider
PARSIPPANY, N.J., Sept. 18, 2024 /PRNewswire/ -- On August 23, 2024, Bloomage released its mid-year financial report, reporting a revenue ofRMB 2.811 billion (~USD 385 million) for the first half of the year. This success reflects the company's strategic transformation from a traditional raw mate...
Kintara Therapeutics Reminds Stockholders to Vote by Thursday to Allow for Completion of the Proposed Merger with TuHURA Biosciences
* Stockholders must vote by 11:59 p.m. ET on September 19, 2024 for their vote to count * A "FOR" vote on Proposals 3 & 5 by holders of a majority of the voting power of Kintara's outstanding shares as ofAugust 14, 2024 is required to allow for completion the proposed merger with TuHURA Biosc...
Week's Top Stories
Most Reposted
UFO SIM Launches in Australia, The Ultimate Travel eSIM Starting at Just AU$2/GB
[Picked up by 310 media titles]
2026-04-01 11:03Sustainable Tourism Impact Fund Expands Investments Across Southeast Asia
[Picked up by 306 media titles]
2026-04-03 11:30Surfin Meta Digital Technology signs MOU with the Philippine Social Security System to explore digital financial service enhancements
[Picked up by 305 media titles]
2026-04-01 09:00From Doomscroll to Page-Turner: BookXcess Launches The Brain Un-Rot Library to Help Rebuild a Generation's Focus
[Picked up by 302 media titles]
2026-03-31 13:53DayOne Announces RM28+ Billion Commitment and Talent Initiatives in Malaysia at Inaugural Tech & AI Career Expo
[Picked up by 291 media titles]
2026-04-01 22:23