Pharmaceuticals
Sanyou Bio Collaborates on ADC Drug Development Entering Phase I Clinical Trials
SHANGHAI, July 16, 2024 /PRNewswire/ -- Sanyou Biopharmaceuticals (Shanghai) Co., Ltd. (hereinafter referred to as "Sanyou Bio") has recently announced a significant milestone in collaboration with Hangzhou Zhongmei Huadong Pharmaceutical Co., Ltd., a wholly-owned subsidiary of Huadong Medicine C...
Biosyngen's BRG01 enters Phase II clinical trial, a first-in-kind autologous EBV-Specific CAR-T Therapy for Solid Tumors on Recurrent/Metastatic Nasopharyngeal Carcinoma
SINGAPORE, July 16, 2024 /PRNewswire/ -- Biosyngen, a leading biotechnology company focused on the development of innovative cell therapies, recently announced that the Center for Drug Evaluation (CDE) of the National Medical Products Administration (NMPA) inChina has approved the initiation of a...
Mabwell Receives NMPA Approval for Clinical Trial of Novel Nectin-4 Targeting ADC in TNBC
SHANGHAI, July 15, 2024 /PRNewswire/ -- Mabwell (688062.SH), an innovative biopharmaceutical company with entire industry chain, announced its novel Nectin-4 targeting ADC (R&D code: 9MW2821) has been approved by the NMPA to enter Phase II clinical trial as monotherapy or in combination with a PD...
HanAll Biopharma Appoints Christopher W Slavinsky as Chief Business Development and Legal Officer
ROCKVILLE, Md. and SEOUL, South Korea, July 15, 2024 /PRNewswire/ -- HanAll
Biopharma (KRX: 009420.KS) announced today the appointment ofChristopher W.
Slavinsky as Chief Business Development and Legal Officer.
CPHI & PMEC Shenzhen 2024: Fostering New Opportunities in Asia and China's Greater Bay Area
SHANGHAI, July 15, 2024 /PRNewswire/ -- CPHI & PMEC China 2024 concluded in June with an exceptional turnout of visitors, setting a new attendance record. Notably, the number of international visitors grew to 11,563, and professionals from the end-to-end pharmaceutical supply chain made meaningfu...
Everest Medicines Announces Positive Topline Results from Maintenance Period of Etrasimod Phase 3 Clinical Trial Conducted in Asia for the Treatment of Moderately-to-Severely Active Ulcerative Colitis
SHANGHAI, July 15, 2024 /PRNewswire/ -- Everest Medicines (HKEX 1952.HK, "Everest", or the "Company"), a biopharmaceutical company focused on the discovery, clinical development, manufacturing and commercialization of innovative therapeutics, today announced positive topline data results of the ...
Basecare Medical (2170. HK) revenue growth exceeds 50% in 2024H1
SUZHOU, China , July 15, 2024 /PRNewswire/ -- Basecare Medical (2170. HK) released an unaudited interim performance forecast onJuly 12th, stating that the company's revenue for the first half of the year was approximatelyRMB 127 million to RMB 138 million, an increase of approximately 49% to 62% ...
Sirnaomics Announces Completion of IND-Enabling Studies of Safety and Efficacy for STP125G with NHP Models, Targeting ApoC3 for Treatment of Cardiovascular Diseases
HONG KONG, GERMANTOWN, Md. and SUZHOU, China, July 12, 2024 /PRNewswire/ -- Sirnaomics Ltd.(the "Company", Stock Code: 2257.HK, together with its subsidiaries, the "Group" or "Sirnaomics"), a leading biopharmaceutical company engaging in discovery and development of advanced RNAi therapeutics, an...
First Patient Dosed in Phase I Clinical Trial of YOLT-201
SHANGHAI, July 11, 2024 /PRNewswire/ -- YolTech Therapeutics, a biotech company developing in vivo gene editing therapies to treat rare genetic diseases, announced today the successful enrollment of the first patient in the Phase I clinical trial of YOLT-201, its independently developed in vivo g...
Sanyou Bio and BioGeometry join forces to create a next-generation Antibody Drug Discovery Platform driven by AIGC
SHANGHAI, July 11, 2024 /PRNewswire/ -- Recently, BioGeometry and Sanyou Bio jointly announced the signing of a strategic partnership agreement. BioGeometry is a digital biology pioneer company that specializes in AI-driven protein design and R&D platform. Sanyou Bio is a world-leading high-tech...
Initiation of cell therapy research and development program for adult T-Cell leukemia / lymphoma (ATLL)
Initiation of cell therapy research and development program for adult T-Cell leukemia / lymphoma (ATLL) TOKYO, July 11, 2024 /PRNewswire/ -- The National Cancer Center (Tokyo, Japan ) and TheUniversity of Pennsylvania (Philadelphia, PA, USA) have licensed patent rights directed to a chimeric...
ADM Korea Announces Niclosamide-based Metabolic Anticancer Drug's First Clinical Trial Target as 'Prostate Cancer Patients Resistant to Hormone Therapy'
* IND for clinical study for combination therapy with hormone therapy in prostate cancer patients to be submitted in August, 2024. * Niclosamide-based metabolic anticancer drug aims to block signaling pathways that allow cancer cells to evade anticancer effects, potentially solving the issue ...
Telix Welcomes CMS Proposal to Improve Payment for Specialised Diagnostic Radiopharmaceuticals
MELBOURNE, Australia, July 11, 2024 /PRNewswire/ -- Telix Pharmaceuticals Limited (ASX: TLX, Telix, the Company) today welcomes proposed changes announced by the Centers for Medicare & Medicaid Services (CMS) for the Hospital Outpatient Prospective Payment System (OPPS) rule to improve payments ...
Eligo Publishes in Nature a Landmark Study That Unlocks Genome Editing of Bacteria in the Gut
PARIS, July 10, 2024 /PRNewswire/ -- For the first time, a team of scientists at Eligo has demonstrated it was possible to genetically modify bacteria with nearly 100% efficiency directly in the gut of animals. This work provides scientists with a novel strategy to better understand how genes fro...
WuXi Biologics Named Constituent of the FTSE4Good Index Series for Fourth Year
SHANGHAI, July 10, 2024 /PRNewswire/ -- WuXi Biologics ("WuXi Bio") (2269.HK), a leading global Contract Research, Development and Manufacturing Organization (CRDMO), announced it has again been named as a constituent company of the FTSE4Good Index Series. This marks the fourth consecutive year t...
Porton J-STAR and Enabling Technologies Consortium Collaboration on Co-processing Platform Technology Development
CHONGQING, China, July 8, 2024 /PRNewswire/ -- Porton J-STAR today announced
that Enabling Technologies Consortium (ETC) has awarded a two-year contract to
J-STAR to collaborate on "Co-processing Platform Technology."
Cambrex to Expand Stability Storage Business with New Facility in Durham, North Carolina
EAST RUTHERFORD, N.J., July 8, 2024 /PRNewswire/ -- Cambrex today announced the expansion of its stability storage business, Q1 Scientific, which offers environmentally-controlled stability storage services to the pharmaceutical, medical device, and life sciences industries. Q1 Scientific will op...
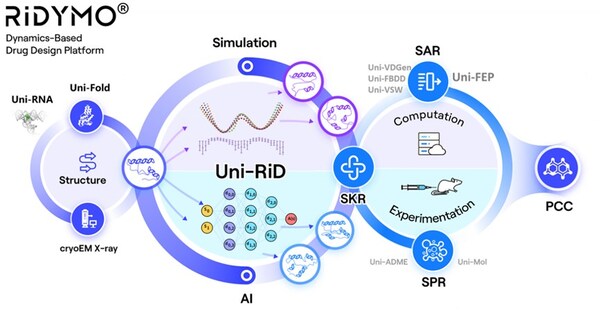
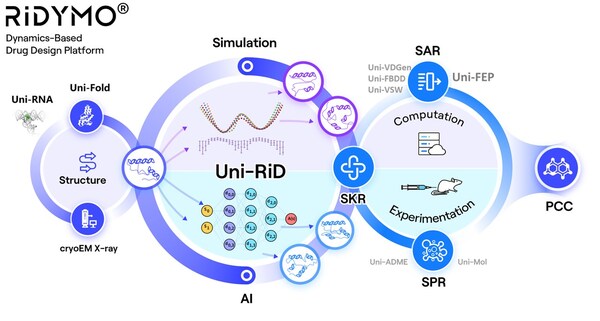
DP Technology Announces Nomination of Development Candidate, a CNS Penetrable Lp-PLA2 Inhibitor for Alzheimer's Disease
BEIJING, July 8, 2024 /PRNewswire/ -- DP Technology, an "AI for Science" paradigm-driven company, today announced the nomination of DPT0416, a novel CNS penetrable small molecule targeting Lp-PLA2, as a preclinical candidate for the treatment of Alzheimer's disease (AD). AD is the most frequent ...
DP Technology Announces Nomination of Development Candidate, a Potential Best-in-Class Lp-PLA2 Inhibitor for DR and DME
* The nomination of novel Lp-PLA2 inhibitor DPT0415 for the treatment of DR & DME. * DPT0415 is a highly potent, selective and safe Lp-PLA2 inhibitor with sufficient target engagement at a dose of 0.3 mpk and robust efficacy in the STZ-induced rat DR model. * This project further validates ...
Immorna Biotherapeutics Receives U.S. FDA IND Clearance to Conduct Phase 1/2 Study of JCXH-211 IV as Monotherapy and in Combination with Checkpoint Inhibitor in Patients with Advanced Solid Tumors
MORRISVILLE, NC., July 8, 2024 /PRNewswire/ -- Immorna Biotherapeutics, Inc. (Immorna), a clinical stage biotechnology company developing both self-replicating and conventional mRNA-based therapeutics and vaccines, announced today that the U.S. Food and Drug Administration (FDA) has cleared its ...
Week's Top Stories
Most Reposted
Tetra Pak Opens Product Development Centre in Thailand, Unlocking A World of Possibilities for Food and Beverage Innovators Across Asia Pacific
[Picked up by 314 media titles]
2026-03-27 11:15Visa Working Capital Index: Asia Pacific CFOs Call for Flexible, Digital Finance Solutions
[Picked up by 309 media titles]
2026-03-24 14:00Indonesia's P2P Lending Grows 25%, JULO Strengthens Credit Quality
[Picked up by 300 media titles]
2026-03-26 18:18ART CENTRAL COMMENCES DISCOVERY-LED ELEVENTH EDITION
[Picked up by 277 media titles]
2026-03-25 16:51RoboSense Beat Expectations, Achieved First-Ever Quarterly Profit on Strong Robotics Growth
[Picked up by 269 media titles]
2026-03-25 22:51