Pharmaceuticals
MediLink Announces Global Clinical Trial Collaboration and Supply Agreement on YL201 Combination Therapy
SUZHOU, China, Oct. 7, 2024 /PRNewswire/ -- MediLink Therapeutics (Suzhou) Co., Ltd. ("MediLink"), a clinical-stage biotech company, today announced a global clinical trial collaboration and supply agreement with Amgen Inc. Amgen will lead a global clinical study to evaluate the therapeutic poten...
TiumBio Announces First Patient Dosed in Phase 2 Clinical Trial of Oral Immuno-Oncology Drug TU2218
* TU2218 is a potentially first-in-class dual inhibitor targeting transforming growth factor beta receptor 1 (TGFR1) and vascular endothelial growth factor receptor 2 (VEGFR2) * The first patient with head and neck squamous cell carcinoma was dosed in the Phase 2a clinical trial of TU2218 *...
WELLlife COVID-19/Influenza A&B Home Test on Sale for Amazon Prime Day
Standfirst: Welllife, a Flu home test in addition to COVID-19 BOLINGBROOK, Ill., Oct. 3, 2024 /PRNewswire/ -- WELLlife, a leading innovator in medical diagnostic and testing solutions, is increasing accessibility to respiratory illness testing this flu season. The WELLlife COVID-19/Influenza A&B...
Doer Biologics Announces First Patient Dosed in Phase 2 Study of DR10624 for Treatment of Severe Hypertriglyceridemia
HANGZHOU, China, Sept. 30, 2024 /PRNewswire/ -- Zhejiang Doer Biologics Co., Ltd. ("Doer Bio"), a clinical stage biopharmaceutical company developing innovative biotherapeutics for metabolic diseases and cancers, today announces that DR10624, its first-in-class (FIC), tri-specific agonist targeti...
IASO Bio Scientist and Leading Chinese Physicians in Onco-Hematology Visit Hematology Center at the State University of Campinas, Brazil, to Explore Cooperation in Cancer Treatment
SHANGHAI and NANJING, China and SAN JOSE, Calif., Sept. 30, 2024 /PRNewswire/ -- OnSeptember 23, in Rio de Janeiro, on the eve of the International Myeloma Society (IMS) Annual Meeting, Dr.Yongke Zhang, Chief Scientific Officer of IASO Biotherapeutics (IASO Bio), along with leading Chinese onco-h...
Akeso's Cadonilimab Receives Second Indication Approval from NMPA for First-Line Treatment of Gastric/GEJ Cancer in All-Comers Population
HONG KONG, Sept. 30, 2024 /PRNewswire/ -- September 30, 2024, Akeso (9926. HK) announced that its internally developed PD-1/CTLA-4 bispecific antibody, cadonilimab, has received approval from the National Medical Products Administration (NMPA) for a new indication: cadonilimab in combination with...
Regor Enters into a Definitive Purchase Agreement for Genentech to Acquire Regor's Portfolio of next-generation CDK inhibitors for the Treatment of Breast Cancer
* Genentech will acquire Regor's next-generation CDK inhibitors for an upfront payment of$850 million in cash. Regor is also eligible to receive additional cash payments contingent on achieving future development, regulatory and commercial milestones CAMBRIDGE, Mass., Sept. 30, 2024 /PRNewswire...
SeromYx Systems and ACROBiosystems Announce Strategic Collaboration on Comprehensive Functional Profiling of Anti-CD20 Monoclonal Antibodies
NEWARK, Del., Sept. 29, 2024 /PRNewswire/ -- SeromYx Systems, a cutting-edge immunology technology company, and ACROBiosystems, an innovative provider for life science solutions and tools, are excited to announce the release of their joint study on the comprehensive functional profiling of approv...
IASO Bio Presented Comparative Clinical Outcomes on the Optimal Lymphodepletion Prior to Infution of Equecabtagene Autoleucel(FucasoTM)in Patients with Relapsed Refractory Multiple Myeloma at 2024 IMS Annual Meeting
SHANGHAI and NANJING, China and SAN JOSE, Calif, Sept. 29, 2024 /PRNewswire/ -- IASO Biotherapeutics ("IASO Bio"), a biopharmaceutical company dedicated to discovering, developing, manufacturing and commercializing innovative cell therapy and antibody products, today announced a poster presentati...
New Beginnings, New Journey | Sanyou Opening of New 10,000-sqm Facility, Ushering in a New Era of Innovation for Bio Drugs
SHANGHAI, Sept. 28, 2024 /PRNewswire/ -- Sanyou Bio recently celebrated a prestigious move into its new 10,000-square-meter R&D building atShanghai headquarters. This is not just a change in physical location, but a qualitative leap in the company infrastructure. It marks a solid step forward for...
111, Inc. Announces Receipt of Notification from Nasdaq
SHANGHAI, Sept. 27, 2024 /PRNewswire/ -- 111, Inc. ("111" or the "Company") (NASDAQ: YI), a leading tech-enabled healthcare platform company committed to reshaping the value chain of healthcare industry by digitally empowering the upstream and downstream inChina, today announced it has received a...
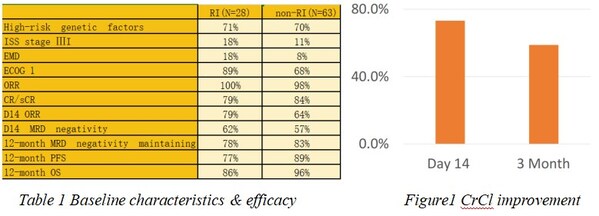
IASO Bio Presented the Outcomes of Relapsed/Refractory Multiple Myeloma (R/RMM) Patients with Renal Impairment Treated with Equecabtagene Autoleucel (Fucaso™) at 2024 IMS Annual Meeting
SHANGHAI and NANJING, China and SAN JOSE, Calif., Sept. 27, 2024 /PRNewswire/ -- IASO Biotherapeutics ("IASO Bio"), a biopharmaceutical company dedicated to discovering, developing, manufacturing and commercializing innovative cell therapy and antibody products, today announced a poster presentat...
Breakthrough Therapy Designation (BTD) for BioCity's SC0062, a selective endothelin type A (ETA) antagonist, granted by National Medical Products Administration for IgA nephropathy (IgAN) with proteinuria
SHANGHAI, Sept. 26, 2024 /PRNewswire/ -- BioCity Biopharma (BioCity) announced its endothelin receptor type A (ETA) selective antagonist SC0062 has been granted the Breakthrough Therapy Designation (BTD) by National Medical Products Administration (NMPA) for the treatment of IgA nephropathy (IgAN...
The World First Liquid Formulation of Recombinant Botulinum Toxin Type A Has Obtained IND Approval by the FDA
CHONGQING, China, Sept. 25, 2024 /PRNewswire/ -- MingMed Biotechnology, an innovative company focused on the in-house discovery and development of novel drugs, recently announced that its partially owned subsidiary, Claruvis Pharmaceutical Co., has received approval from the U.S. Food and Drug A...
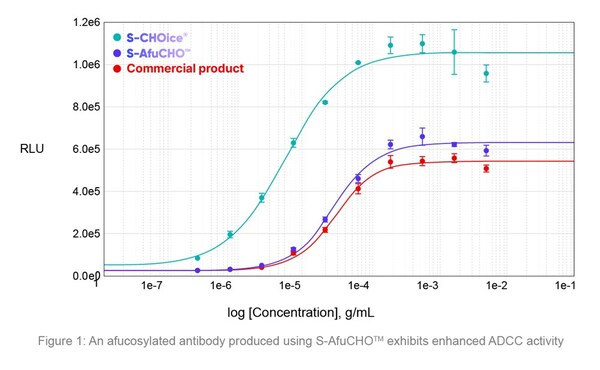
Samsung Biologics launches development platforms for enhanced therapeutic efficacy
* Samsung Biologics showcases new innovative development platforms – S-AfuCHO TM and S-OptiChargeTM – at BioProcess International 2024 * New technology platforms to proactively address evolving industry trends and enable high-quality development INCHEON, South Korea, Sept. 25, 2024 /PRNewswire...
XtalPi Launches Computational Chemistry Software for Drug Discovery: XMolGen and XFEP
BOSTON and SHANGHAI, Sept. 24, 2024 /PRNewswire/ -- Today, XtalPi announced the official launch of XFEP and XMolGen, two proprietary software products designed to accelerate and enhance the efficiency of drug discovery. Upon this launch, XtalPi will offer commercial licenses with flexible terms f...
LongBio announced the completion of Series B2 financing, led by Qiming Venture Partners
SHANGHAI, Sept. 24, 2024 /PRNewswire/ -- LongBio Pharma (Suzhou) Co., Ltd. ("LongBio" or the "Company"), a Phase III-stage biotech company focused on the R&D of innovative antibody and fusion protein drugs for allergies and complement-mediated diseases, announced the completion of its Series B2 ...
Nona Biosciences Enters Strategic Collaboration with Alkyon Therapeutics for Next-Generation Immunotherapeutics Discovery
CAMBRIDGE, Mass., Sept. 24, 2024 /PRNewswire/ -- Nona Biosciences, a global biotechnology company providing a total solution from "Idea to IND" (I to I™ ), announced today that it has entered into a strategic collaboration with Alkyon Therapeutics, Inc. (AlkyonTx), aSan Diego-based biotechnology ...
Impressive preliminary objective response rates (ORR) of 36% in NSCLC and 80% in EGFRmut NSCLC: Interim Results of a Phase 1 Study of BC3195, a First-in-Class ADC Targeting CDH3, Presented by BioCity at ESMO 2024
SHANGHAI, Sept. 23, 2024 /PRNewswire/ -- On September 14, 2024, BioCity presented interim clinical results on the safety and efficacy of its first-in-class antibody-drug conjugate (ADC) BC3195 targeting CDH3 (P-Cadherin) in a Phase I clinical trial at the European Society for Medical Oncology (ES...
WuXi Biologics Included in Hang Seng ESG 50 Index
HONG KONG, Sept. 23, 2024 /PRNewswire/ -- WuXi Biologics ("WuXi Bio") (2269.HK), a leading global Contract Research, Development and Manufacturing Organization (CRDMO), today announced that it has been included in the Hang Seng ESG 50 Index, making it the only pharmaceutical company selected this...
Week's Top Stories
Most Reposted
Visa partners with Laufey to spread the magic of travel in Asia Pacific; to be Official Payment Partner for Laufey: A Matter of Time Tour
[Picked up by 309 media titles]
2026-03-04 12:35Klook's Spring Readiness Index shows how Asia's travelers are preparing for spring travel across Japan, South Korea, and Mainland China
[Picked up by 291 media titles]
2026-03-03 15:49COL and NASDAQ-Listed BeLive Holdings Unveil World's First "Microdrama in a Box" in Headline Hong Kong FILMART 2026 Launch
[Picked up by 287 media titles]
2026-03-05 17:14Gene Solutions Announces Strategic Entry into Türkiye with Robust Real-World Evidence at the 2nd International Hereditary Cancers Congress 2026
[Picked up by 267 media titles]
2026-03-04 10:00The Ministry of Finance of Thailand, the Bank of Thailand, and the International Monetary Fund Underscore Significant Progress Toward the 2026 IMF-World Bank Group Annual Meetings
[Picked up by 266 media titles]
2026-03-05 12:18